Cholangitis, Primary Sclerosing
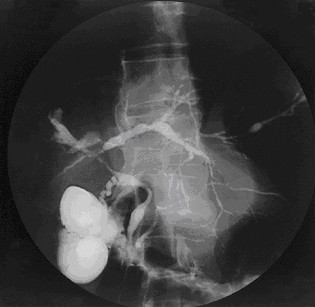
Description
Primary sclerosing cholangitis (PSC) is a slowly progressive cholestatic liver disease characterized by fibroobliterative inflammation of the biliary tract, leading to cirrhosis and portal hypertension. It is a major indication for liver transplantation (Sheth et al., 2003).
Approximately 75 to 80% of PSC cases are associated with inflammatory bowel disease (IBD; see 266600), and 2.5 to 7.5% of patients with IBD develop PSC (Lee and Kaplan, 1995).
MappingIn a genomewide association study of in 715 Scandinavian and German individuals with PSC and 2,962 controls, Melum et al. (2011) found the strongest associations with SNPs in the HLA complex at chromosome 6p21, peaking at rs3134792 in HLA-B (142830) (p = 6.8 x 10(-49)). Carriers of the G allele at rs3134792 were HLA-B*08 carriers in 99% of the cases, and were HLA-DRB1*03 (142857) carriers in 90% of the cases. Inclusion of this SNP as a covariate showed a complex residual association signal in the vicinity of the HLA class II region (p = 7.6 x 10(-17) for rs9272723), suggesting the presence of multiple causative loci within the region.
Genetic Heterogeneity
In a genomewide association study of in 715 Scandinavian and German individuals with PSC and 2,962 controls, Melum et al. (2011) also detected a significant association with rs3197999 in the MST1 gene (142408) on chromosome 3p21 (p = 1.4 x 10(-9); odds ratio (OR) of 1.51). In a replication study of 1,025 PSC cases and 2,174 controls, there was again a significant association between PSC and MST1 (combined p = 1.1 x 10(-16)). In addition, the most prominent association was with a nonsynonymous SNP (arg689-to-cys; R689C) (rs3197999) in MST1. This SNP has also been associated with IBD in a study of 1,765 IBD patients (p = 3.62 x 10(-6)) (Goyette et al., 2008). The findings suggested that the MST1 locus represents an important overlapping susceptibility locus for PSC and IBD: see IBD12 (612241). In the same cohorts, Melum et al. (2011) also found a significant association with rs6720394 near the BCL2L11 gene (603827) at chromosome 2q13 (combined p = 4.1 x 10(-8)).
PathogenesisUsing flow cytometry, Eksteen et al. (2004) found substantially increased expression of CCR9 (604738) on liver-infiltrating lymphocytes (LILs) in patients with primary sclerosing cholangitis compared with organ donor controls and patients with other chronic inflammatory liver diseases. Immunohistochemical and Western blot analyses demonstrated that CCL25 (602565), which is normally expressed in thymus and intestine only, was highly expressed in livers of PSC patients but not other liver disease patients. In vitro, CCR9-positive LILs preferentially migrated to CCL25 and could be triggered by CCL25 to bind immobilized MADCAM1 (102670) via alpha-4/beta-7 integrins (see 147559). Eksteen et al. (2004) proposed that T cells activated in the gut during active inflammatory bowel disease differentiate into effector cells with the ability to bind to both hepatic and mucosal endothelium. They suggested that CCL25 and MADCAM1 cooperate in recruiting mucosal CCR9-positive lymphocytes to the liver in PSC.
Sheth et al. (2003) hypothesized that dysfunction of CFTR (602421) may explain why a subset of patients with IBD develop PSC. They prospectively evaluated CFTR genotype and phenotype in 19 patients with PSC compared with 18 patients with IBD and no liver disease, 17 with primary biliary cirrhosis, 81 with cystic fibrosis (CF; 219700), and 51 healthy controls. They found an increased prevalence of CFTR abnormalities in heterozygous state in PSC as demonstrated by molecular and functional analyses, and concluded that these abnormalities may contribute to the development of PSC in a subset of patients with IBD. Eighty-nine percent of PSC patients carried genotypes containing the 1540G variant (602421.0023) resulting in decreased functional CFTR compared with 57% of disease controls (p = 0.03). Only 1 of 19 PSC patients had neither a CFTR mutation nor the 1540G variant. CFTR chloride channel function assessed by nasal potential difference testing demonstrated a reduced median isoproterenol response in PSC patients compared with disease controls and healthy controls.
Takeda et al. (2008) found that administration of a monoclonal anti-Dr5 (603612) antibody specifically induced cholestatic liver failure and apoptosis of cholangiocytes in B6 mice, but not other strains of mice. Trail (603598)- or Dr5-null B6 mice were relatively resistant to common bile duct ligation-induced cholestasis. In addition, common bile duct ligation augmented Dr5 expression in cholangiocytes of wildtype mice, sensitizing them to Dr5-mediated cholangitis. Histologic appearance of mouse liver samples was reminiscent of human primary sclerosing cholangitis. Cholangiocytes derived from patients with PSC showed increased expression of DR5 and TRAIL compared to controls. Apoptosis of cholangiocytes was also observed in human samples. The findings suggested a role for TRIAL/DR5-mediated apoptosis in cirrhotic biliary diseases.