Neural Tube Defects, Susceptibility To
A number sign (#) is used with this entry because of evidence that susceptibility to the development of neural tube defects (NTDs) is conferred by variation in the VANGL1 (610132), VANGL2 (600533), CELSR1 (604523), or FUZ (610622) genes. An association has been reported with variants in the T locus (601397) on chromosome 6q.
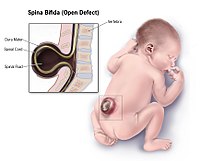
DescriptionNeural tube defects are the second most common type of birth defect after congenital heart defects. The 2 most common NTDs are open spina bifida, also known as spina bifida cystica (SBC) or myelomeningocele, and anencephaly (206500) (Detrait et al., 2005). Spina bifida occulta (SBO), a bony defect of the spine covered by normal skin, is a mild form of spina bifida that is often asymptomatic. The term 'spinal dysraphia' refers to both SBC and SBO (Botto et al., 1999; Fineman et al., 1982). The most severe neural tube defect, craniorachischisis (CRN), leaves the neural tube open from the midbrain or rostral hindbrain to the base of the spine (summary by Robinson et al., 2012).
Neural tube defects represent a complex trait with multifactorial etiology encompassing both genetic and environmental components (summary by Bartsch et al., 2012 and Lei et al., 2014).
An X-linked form of spina bifida has been suggested; see 301410. See also folate-sensitive neural tube defects (601634), which are caused by genes involved in folate metabolism.
Clinical FeaturesFineman et al. (1982) studied 4 families in which multiple members had spina bifida cystica and/or spina bifida occulta. Excluding the probands, the frequency of all types of spinal/vertebral defects in family members was found to be 30/58 (52%). Statistical analysis indicated autosomal dominant inheritance with a penetrance of 75%. The authors concluded that both forms of spina bifida are different expressions of the same dominant gene in these kindreds. Spina bifida occulta was present in 15% of 100 control individuals.
Fellous et al. (1982) reported a 5-generation family with spina bifida associated with sacral agenesis (see 600145). Abnormalities ranged from complete absence of the sacrum, with or without spina bifida aperta, to spina bifida occulta. The condition appeared in a man with 4 children who were all affected, and thereafter, to varying degrees, in 17 of his 28 descendants. The authors suggested autosomal dominant inheritance. Linkage analysis suggested a locus on chromosome 6 loci near PGM3 (172100) (lod score = 1.85 at a recombination fraction of 0.087).
Nickel et al. (1994) reported 3 unrelated patients with 22q11 deletion syndrome, 2 with VCFS (192430) and 1 with DiGeorge syndrome (188400), who also had neural tube defects. However, in a follow-up study of 295 patients with spina bifida, Nickel and Magenis (1996) concluded that 22q11 deletion is an infrequent cause of NTD.
Reviews
Botto et al. (1999) and Detrait et al. (2005) provided reviews of neural tube defects. De Marco et al. (2006) provided a detailed review of neurulation and the possible etiologies of neural tube defects.
Other FeaturesChiari malformation type II (CM2; 207950), also known as the Arnold-Chiari malformation, is often associated with open spina bifida. Lindenberg and Walker (1971) described the Arnold-Chiari malformation in 2 successively born daughters of nonconsanguineous parents. Both children had associated hydrocephalus and lumbar meningomyelocele.
InheritanceNeural tube defects represent a complex trait with multifactorial etiology encompassing both genetic and environmental components (summary by Bartsch et al., 2012 and Lei et al., 2014).
Record and McKeown (1950) estimated a recurrence risk of spina bifida in sibs of affected children to be 4%. Lorber (1965) reported familial occurrence of spina bifida. He suggested recessive inheritance with reduced penetrance and estimated the risk of recurrence of spina bifida cystica, anencephaly, or hydrocephalus in subsequently born offspring to be about 8%. In England, Carter and Roberts (1967) estimated the risk of a third child having spina bifida or anencephaly, 2 having previously been affected, to be about 1 in 10. Lorber and Levick (1967) found spina bifida occulta in 14.3% of 188 mothers and 26.8% of 179 fathers of patients with spina bifida cystica, and in 5% of 200 controls. However, spina bifida occulta was not more common among parents with more than 1 child with spina bifida cystica and neither parent had it in the majority of families.
Carter et al. (1976) found as high a frequency of anencephaly and spina bifida cystica among the sibs of patients with spinal dysraphism as among the sibs with other open neural tube defects, suggesting a common etiology of various spinal defects.
MappingFrom studies in a large kindred, Amos et al. (1975) concluded that there may be a gene locus for spina bifida occulta linked to the HLA complex on chromosome 6. Early interest in this possibility was stimulated by the T region of the mouse (601397), which determines morphogenesis, especially of the tail, and is linked to the H-2 region on mouse chromosome 17. Mendell et al. (1979) studied 2 extensive North Carolina kindreds suggesting that spina bifida occulta and/or asymmetry of the facet joints was determined by a gene at a locus linked to HLA. The total lod score was 2.21 at theta = 0.05.
Bobrow et al. (1975) and de Bruyere et al. (1977) found no linkage of spina bifida with HLA. Vannier et al. (1981) found a high frequency of spina bifida occulta and lateral asymmetry of the lumbosacral spine in sibs and parents. However, Vannier et al. (1981) found no association between these abnormalities and the HLA system. Jorde (1983) concluded that linkage to 6p markers could be excluded.
Erickson (1988) reviewed arguments for the assertion that there is no HLA-linked equivalent of the t-complex of the mouse. He emphasized that the essential attributes of the murine t-complex are distortion of prezygotic transmission ratios and suppression of crossover rather than developmental recessive mutations leading to embryonic arrest.
Molecular GeneticsVariation At The T Locus
Morrison et al. (1996) reported that an allelic variant of the T locus, referred to as TIVS7-2 (601397.0001), showed a bias in transmission from heterozygous parents to offspring with neural tube defects in Dutch and U.K. families. Shields et al. (2000) also found an association between the TIVS7-2 allele and neural tube defects. However, Trembath et al. (1999) and Speer et al. (2002) found no association.
Jensen et al. (2004) observed that individuals carrying one or more copies of the TIVS7-2 allele have a 1.6-fold increased risk of spina bifida compared with individuals with 0 copies.
Mutations in the VANGL1 Gene
In a study of 144 patients with neural tube defects, Kibar et al. (2007) found mutations in the VANGL1 gene (610132.0001-610132.0003) in 3 patients, 2 with a familial and 1 with a sporadic form of the disorder. The mutations were not found among 106 ancestrally matched controls or 65 control samples obtained from CEPH (Centre d'Etude du Polymorphisme Humain). The findings implicated variation in VANGL1, a human homolog of a Drosophila gene involved in establishing planar cell polarity, as a risk factor in neural tube defects.
In 3 (2.1%) of 144 unrelated patients with various forms of neural tube defects from Slovakia, Romania, and Germany who underwent direct sequencing of the VANGL1 gene, Bartsch et al. (2012) identified 3 different heterozygous missense variants (R173H, R186H, and G25R). The patients had sporadic neural tube defects, sporadic spinal lipoma and tethered cord, and sporadic lumbosacral meningomyelocele, respectively. All variants occurred at highly conserved residues and were not found in SNP databases or in 357 controls; however, functional studies were not performed. At least 1 unaffected relative of 1 of the patients also carried the variant.
By direct sequencing of the VANGL1 gene in 53 patients with various neural tube defects and 27 couples with an affected pregnancy, Merello et al. (2015) identified 3 heterozygous missense variants (A187V, D389H, and R517H) that were not present in 200 matched Italian controls. The variants were found in a girl with lipomyeloschisis, an unaffected woman who had a pregnancy complicated by anencephaly, and a boy with myelomeningocele, respectively. The 2 children each inherited the variant from an unaffected parent. Two additional synonymous variants that were predicted to cause splice site changes were found in a child with cephalocele and in the father of a fetus with myelomeningocele. Functional studies of the variants were not performed. In a review of the literature, Merello et al. (2015) found a significant association between rare variants in the VANGL1 gene and neural tube defects (odds ratio of 2.24; p = 7.0 x 10(-5)). The finding suggested that variation in the VANGL1 gene may play a role as a genetic risk factor for neural tube defects, although likely only for a minority of cases.
Mutations in the VANGL2 Gene
Lei et al. (2010) identified 3 different heterozygous missense mutations in the VANGL2 gene (see, e.g., 600533.0001 and 600533.0002) in 3 of 163 unrelated Chinese Han stillborn or miscarried fetuses with neural tube defects, including anencephaly. The authors postulated that loss of function defects in this gene have a lethal effect during in utero development in humans, and noted that mouse studies have indicated the loss of Vangl2 results in defects in neural tube closure.
Kibar et al. (2011) sequenced the VANGL2 gene in a population-based study of 673 patients with various forms of neural tube defects. Six potentially pathogenic heterozygous missense mutations were identified in 7 patients, including 3 at positions that were absolutely conserved through zebrafish and Drosophila (R135W, R177H, and R270H), and 3 at positions that were highly conserved (L242V, T247M, and R482H). Two patients had open NTDs with myelomeningocele and 5 had closed NTDs, which was a statistically significant difference (p = 0.027). However, 2 unaffected parents carried 2 of the mutations, and another mutation (R105C) was found in 1 of 287 controls. Functional studies of the mutations were not performed. Kibar et al. (2011) suggested that variation in the VANGL2 gene may predispose to neural tube defects, but noted that the findings needed to be confirmed.
Merello et al. (2015) did not find an association between rare variants in the VANGL2 gene and neural tube defects among 879 Italian patients who underwent sequencing.
Mutations in the FUZ Gene
Seo et al. (2011) found 5 heterozygous missense mutations in the FUZ gene (see, e.g., 610622.0001-610622.0003) among 234 Italian patients with neural tube defects. These sequence variants were absent in 130 matching Italian controls and in over 250 controls sequenced as part of the 1000 Genomes Project. One mutation was found to interfere with ciliogenesis in 2 independent assays but did not affect cell directional movement. Another was found to have a striking effect on directional cell movement without affecting ciliogenesis, and a third mutation was found to affect both functions.
Mutations in the CELSR1 Gene
By direct sequencing of the CELSR1 gene, Lei et al. (2014) identified heterozygous frameshift mutations in the CELSR1 gene (604523.0001 and 604523.0002) in 2 (1%) of 192 patients from California with the neural tube defect spina bifida. In vitro functional analysis showed that both mutations changed subcellular localization of the CELSR1 protein and impaired the physical association between CELSR1 and VANGL2, diminishing the ability to recruit VANGL2 for cell-cell contact. Studies of patient tissue were not performed. Six additional heterozygous missense variants, 4 of which were predicted to be pathogenic (A1023G, I1124M, T1362M, and R2497C), were also found in patients but not in controls or in the Exome Variant Server database; however, functional studies of these variants were not performed.
Exclusion Studies
Volcik et al. (2002) used the transmission disequilibrium test (TDT) to determine if genes in the PAX family play a role in the formation of NTDs; they also performed single-strand conformation analysis (SSCA) and direct sequencing. Although multiple variations were detected in each of the PAX genes with significant TDT results, the authors concluded that it is unlikely that these variations contribute to susceptibility for spina bifida. By analysis of homeobox genes in 459 spina bifida patients and their parents, as well as within gene regions of 8 mouse models that exhibit NTDs, Volcik et al. (2002) obtained no significant findings with the markers tested.
Associations Pending Confirmation
For discussion of a possible association between variation in the PRICKLE1 gene and neural tube defects, see 608500.
For discussion of a possible association between variation in the CCL2 gene and neural tube defects, see 158105.
For discussion of a possible association between variation in the SCRIB gene and neural tube defects, see 607733.
For discussion of a possible association between variation in the DACT1 gene and neural tube defects, see 607861.
For discussion of a possible association between variation in the SHROOM3 gene and neural tube defects, see 604570.0001 and 605470.0002.
Population GeneticsSpina bifida and anencephaly are common birth defects, affecting 0.5 to 2 per 1,000 pregnancies worldwide. Craniorachischisis, the most severe neural tube defect, is considered rare, although estimates of prevalence range from 1 in 100,000 in the United States to 1 in 1,000 in northern China (summary by Robinson et al., 2012).
Animal ModelAdalsteinsson and Basrur (1984) concluded that spina bifida in Icelandic lambs is an autosomal recessive.
Helwig et al. (1995) reported that mice who are doubly heterozygous for the mutants 'undulated' and 'Patch' have a phenotype reminiscent of an extreme form of spina bifida occulta in humans. The unexpected phenotype in double-mutant and not single-mutant mice showed that novel congenital anomalies such as spina bifida can result from interaction between products of independently segregating loci. This is an example of digenic inheritance. (The 'undulated' mutation is related to the mouse Pax1 gene (PAX1; 167411), and the 'Patch' mutation is related to deletion of the mouse Pdgfra gene (PDGFRA; 173490).)
Juriloff and Harris (2000) reviewed the numerous mouse models of NTDs, as well as the zonal pattern of neural tube closure and the effect of maternal nutrients on neural tube closure.