Andersen-Tawil Syndrome
Summary
Clinical characteristics.
Andersen-Tawil syndrome (ATS) is characterized by a triad of: episodic flaccid muscle weakness (i.e., periodic paralysis); ventricular arrhythmias and prolonged QT interval; and anomalies including low-set ears, widely spaced eyes, small mandible, fifth-digit clinodactyly, syndactyly, short stature, and scoliosis. Affected individuals present in the first or second decade with either cardiac symptoms (palpitations and/or syncope) or weakness that occurs spontaneously following prolonged rest or following rest after exertion. Mild permanent weakness is common. Mild learning difficulties and a distinct neurocognitive phenotype (i.e., deficits in executive function and abstract reasoning) have been described.
Diagnosis/testing.
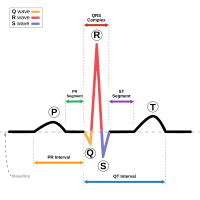
The diagnosis of ATS is established in an individual with characteristic clinical and ECG findings and/or identification of a pathogenic variant in KCNJ2.
Management.
Treatment of manifestations: For episodic weakness: if serum potassium concentration is low (<3.0 mmol/L), administration of oral potassium (20-30 mEq/L) every 15-30 minutes (not to exceed 200 mEq in a 12-hour period) until the serum concentration normalizes; if a relative drop in serum potassium within the normal range causes episodic paralysis, an individual potassium replacement regimen with a goal of maintaining serum potassium levels in the high range of normal can be considered; if serum potassium concentration is high, ingesting carbohydrates may lower serum potassium levels. Mild exercise may shorten or reduce the severity of the attack.
Prevention of primary manifestations: Reduction in frequency and severity of episodic attacks of weakness with lifestyle/dietary modification to avoid known triggers; use of carbonic anhydrase inhibitors; daily use of slow-release potassium supplements; implantable cardioverter-defibrillator for those with tachycardia-induced syncope. Empiric treatment with flecainide should be considered for significant, frequent ventricular arrhythmias in the setting of reduced left ventricular function.
Prevention of secondary complications: Cautious use of antiarrhythmic drugs (particularly class I drugs) that may paradoxically exacerbate the neuromuscular symptoms.
Surveillance: Annual screening of asymptomatic individuals with a KCNJ2 pathogenic variant with a 12-lead ECG and 24-hour Holter monitoring.
Agents/circumstances to avoid: Medications known to prolong QT intervals; salbutamol inhalers (may exacerbate cardiac arrhythmias); thiazide and other potassium-wasting diuretics (may provoke drug-induced hypokalemia and could aggravate the QT interval prolongation).
Evaluation of relatives at risk: Molecular genetic testing if the pathogenic variant is known; if not, detailed neurologic and cardiologic evaluation, 12-lead ECG, and 24-hour Holter monitoring to reduce morbidity and mortality through early diagnosis and treatment of at-risk relatives.
Genetic counseling.
ATS is inherited in an autosomal dominant manner. At least 50% of individuals diagnosed with ATS have an affected parent. Up to 50% of affected individuals have ATS as the result of a de novo pathogenic variant. Each child of an individual with ATS has a 50% chance of inheriting the disorder. Prenatal diagnosis for pregnancies at increased risk is possible if the KCNJ2 pathogenic variant has been identified in an affected family member.
Diagnosis
Suggestive Findings
Andersen-Tawil syndrome (ATS) should be suspected in individuals with either A or B:
- A.
Presence of two of the following three criteria:
- Periodic paralysis
- Symptomatic cardiac arrhythmias or electrocardiographic evidence of enlarged U-waves, ventricular ectopy, or a prolonged QTc or QUc interval
- Characteristic facies, dental anomalies, small hands and feet, AND at least two of the following:
- Low-set ears
- Widely spaced eyes
- Small mandible
- Fifth-digit clinodactyly
- Syndactyly of toes 2 and 3
- B.
One of the above three criteria AND at least one other family member who meets two of the three criteria [Statland et al 2018]
Supportive Findings
Individuals with either episodic weakness or cardiac symptoms require careful evaluation by a neurologist and/or cardiologist as well as measurement of serum potassium concentration (baseline and during attacks of flaccid paralysis), a 12-lead ECG, a 24-hour Holter monitor, and possibly the long exercise protocol.
- Serum potassium concentration during episodes of weakness may be elevated, normal, or, most commonly, reduced (<3.5 mmol/U) [Sansone & Tawil 2007, Statland et al 2018].
- Routine nerve conduction electrophysiology is normal between episodes. A more sensitive electrophysiologic study, the long exercise protocol, may reveal an immediate post-exercise increment followed by an abnormal decrement in the compound motor action potential amplitude (>40%) [Katz et al 1999] or area (>50%) 20-40 minutes post exercise [Kuntzer et al 2000, Fournier et al 2004]. In a study of 11 individuals with ATS, 82% met long-exercise amplitude decrement criteria for abnormal testing [Tan et al 2011].
- Electrocardiogram may reveal characteristic abnormalities including prominent U waves, prolonged Q-U intervals, premature ventricular contractions, polymorphic ventricular tachycardia, and bidirectional ventricular tachycardia [Zhang et al 2005, Delannoy et al 2013, Koppikar et al 2015, Statland et al 2018].
- 24-hour Holter monitoring is important to document the presence, frequency, and duration of ventricular tachycardia (VT) and the presence or absence of associated symptoms.
Establishing the Diagnosis
The diagnosis of ATS is established in a proband with the presence of A or B in Suggestive Findings and/or by identification of a heterozygous pathogenic variant in KCNJ2 by molecular genetic testing (see Table 1).
Molecular genetic testing approaches can include a combination of gene-targeted testing (single-gene testing, multigene panel) and comprehensive genomic testing (exome sequencing, genome sequencing) depending on the phenotype.
Gene-targeted testing requires that the clinician determine which gene(s) are likely involved, whereas genomic testing does not. Because the phenotype of ATS is broad, individuals with the distinctive findings described in Suggestive Findings are likely to be diagnosed using gene-targeted testing (see Option 1), whereas those with a phenotype indistinguishable from many other inherited disorders with cardiac arrhythmias are more likely to be diagnosed using genomic testing (see Option 2).
Option 1
When the phenotypic and laboratory findings suggest the diagnosis of ATS molecular genetic testing approaches can include serial single-gene testing or use of a multigene panel:
- Serial single-gene testing. Sequence analysis detects small intragenic deletions/insertions and missense, nonsense, and splice site variants; typically, exon or whole-gene deletions/duplications are not detected. Perform sequence analysis of KCNJ2 first. If no pathogenic variant is found perform gene-targeted deletion/duplication analysis of KCNJ2 to detect intragenic deletions or duplications. If no KCNJ2 pathogenic variant is found, perform sequence analysis of KCNJ5 (see Table 1, footnote 7).
- A multigene panel that includes KCNJ2, KCNJ5, and other genes of interest (see Differential Diagnosis and Long QT Syndrome) is most likely to identify the genetic cause of the condition at the most reasonable cost while limiting identification of variants of uncertain significance and pathogenic variants in genes that do not explain the underlying phenotype. Note: (1) The genes included in the panel and the diagnostic sensitivity of the testing used for each gene vary by laboratory and are likely to change over time. (2) Some multigene panels may include genes not associated with the condition discussed in this GeneReview. (3) In some laboratories, panel options may include a custom laboratory-designed panel and/or custom phenotype-focused exome analysis that includes genes specified by the clinician. (4) Methods used in a panel may include sequence analysis, deletion/duplication analysis, and/or other non-sequencing-based tests.For an introduction to multigene panels click here. More detailed information for clinicians ordering genetic tests can be found here.
Option 2
When the phenotype is indistinguishable from many other inherited disorders characterized by cardiac arrhythmias, comprehensive genomic testing (which does not require the clinician to determine which gene[s] are likely involved) is the best option. Exome sequencing is most commonly used; genome sequencing is also possible.
For an introduction to comprehensive genomic testing click here. More detailed information for clinicians ordering genomic testing can be found here.
Table 1.
Molecular Genetic Testing Used in Andersen-Tawil Syndrome
| Gene 1 | Test Method | Proportion of Probands with a Pathogenic Variant 2 Detectable by This Method |
|---|---|---|
| KCNJ2 | Sequence analysis 3 | ~70% 4 |
| Gene-targeted deletion/duplication analysis 5 | Unknown 6 | |
| KCNJ5 | Sequence analysis 3 | See footnote 7 |
| Gene-targeted deletion/duplication analysis 5 | Unknown, none reported | |
| Unknown | ~30% |
- 1.
See Table A. Genes and Databases for chromosome locus and protein.
- 2.
See Molecular Genetics for information on allelic variants detected in this gene.
- 3.
Sequence analysis detects variants that are benign, likely benign, of uncertain significance, likely pathogenic, or pathogenic. Pathogenic variants may include small intragenic deletions/insertions and missense, nonsense, and splice site variants; typically, exon or whole-gene deletions/duplications are not detected. For issues to consider in interpretation of sequence analysis results, click here.
- 4.
Plaster et al [2001], Ai et al [2002], Andelfinger et al [2002], Tristani-Firouzi et al [2002], Donaldson et al [2003], Hosaka et al [2003]
- 5.
Gene-targeted deletion/duplication analysis detects intragenic deletions or duplications. Methods used may include: quantitative PCR, long-range PCR, multiplex ligation-dependent probe amplification (MLPA), and a gene-targeted microarray designed to detect single-exon deletions or duplications.
- 6.
Large deletions of KCNJ2 are reported [Lestner et al 2012, Vergult et al 2012, Marquis-Nicholson et al 2014]. The prevalence of large deletions is unknown.
- 7.
One individual with periodic paralysis, ventricular arrhythmias, no history of hypertension, and normal plasma aldosterone levels was found to have a p.Gly387Thr variant in KCNJ5. Of individuals with suspected ATS in whom a KCNJ2 pathogenic variant was not identified, 1/21 had a p.Thr158Ala KCNJ5 variant identified [Kokunai et al 2014].
Clinical Characteristics
Clinical Description
Andersen-Tawil syndrome (ATS) is characterized by a triad of features:
- Episodic flaccid muscle weakness (periodic paralysis)
- Cardiac abnormalities (ventricular arrhythmias, prolonged QTc or QUc intervals, and prominent U waves)
- Distinctive dysmorphic features
Affected individuals present initially with either periodic paralysis or cardiac symptoms (palpitations and/or syncope) in the first or second decade [Tawil et al 1994, Tristani-Firouzi et al 2002]; however, prospective standardized natural history data are not yet available. The phenotype is highly variable. The characteristic triad is present in 58%-78% of individuals with KCNJ2 pathogenic variants [Statland et al 2018].
Weakness. Intermittent weakness occurs spontaneously, or alternatively may be triggered by prolonged rest or rest following exertion. The attack frequency, duration, and severity are variable between and within affected individuals. Mild permanent weakness is common [Tristani-Firouzi et al 2002]. Affected individuals can develop fixed proximal weakness over time.
Ventricular arrhythmias including bidirectional ventricular tachycardia (VT), polymorphic VT, and multifocal premature ventricular contractions may be asymptomatic, or may manifest (most commonly) as palpitations. Less common symptomatic presentations include syncope, cardiac arrest, or sudden death [Andelfinger et al 2002, Tristani-Firouzi et al 2002, Donaldson et al 2003]. While the ECG may reveal a long QTc (LQT) interval, characteristic T-U patterns including enlarged U waves, a wide T-U junction, and prolonged terminal T-wave downslope distinguish ATS from other LQT syndromes [Zhang et al 2005, Haruna et al 2007]. A large case series found no significant difference in the incidence of ventricular tachyarrhythmias between individuals with typical and atypical presentations of ATS [Kimura et al 2012]. A retrospective multicenter French study of 36 individuals with ATS followed for an average of 9.5 years reported no deaths in follow up; four individuals experienced syncope and one individual had a non-fatal cardiac arrest [Delannoy et al 2013].
Dilated cardiomyopathy was observed in two of three affected individuals in a single kindred with pathogenic variant p.Arg218Trp [Schoonderwoerd et al 2006]. Additionally, cardiomyopathy was documented and reversed by treatment with flecainide in an individual with pathogenic variant p.Arg67Trp [Pellizzón et al 2008], and reversed by bisoprolol in another individual with typical clinical features of ATS and novel variant p.Leu222Ser [Rezazadeh et al 2016]. These reports suggest that dilated cardiomyopathy is a secondary phenotype as a consequence of chronic tachycardia rather than a primary phenotypic manifestation. Additional study is needed to further delineate this clinical feature.
Distinctive physical features recognized initially included low-set ears, widely spaced eyes, small mandible, fifth-digit clinodactyly, second and third toe syndactyly, short stature, broad nasal root, and scoliosis [Andersen et al 1971, Tristani-Firouzi et al 2002, Donaldson et al 2003]. Dental enamel discoloration was noted in two kindreds with the p.Gly300Asp and p.Arg218Trp pathogenic variants [Davies et al 2005].
Detailed, prospectively collected data in ten individuals with confirmed KCNJ2 pathogenic variants have expanded the phenotype to include a characteristic facies and dental and skeletal anomalies [Yoon et al 2006a].
- Characteristic facies include broad forehead, short palpebral fissures, wide nasal bridge with bulbous nose, hypoplasia of maxilla and mandible, thin upper lip, and a triangular face.
- Dental findings include (among others) persistent primary dentition, multiple missing teeth (oligodontia), and dental crowding.
- Skeletal findings include mild syndactyly of toes two and three as well as fifth-digit clinodactyly.
- Novel findings include small hands and feet (<10th centile for age) and joint laxity.
Note: Isolated reports of renal anomalies include unilateral hypoplastic kidney [Andelfinger et al 2002] and renal tubular defect [Davies et al 2005].
Mild learning difficulties have been described [Davies et al 2005]. A distinct neurocognitive phenotype (i.e., deficits in executive function and abstract reasoning) has been recognized in individuals with a KCNJ2 pathogenic variant despite IQ levels similar to those of their unaffected sibs [Yoon et al 2006b]. Growth restriction and developmental delay have been described as well [Kim et al 2016].
Afebrile seizures occurring only in infancy were reported in 4/23 (17%) of a Japanese cohort with molecularly confirmed ATS [Haruna et al 2007].
Genotype-Phenotype Correlations
Whether a KCNJ2 pathogenic variant is present or not, individuals with clinically defined ATS are phenotypically indistinguishable [Tristani-Firouzi et al 2002, Donaldson et al 2003].
In a case series that evaluated for KCNJ2 pathogenic variants in individuals with typical (>2 ATS features) and atypical (only 1 ATS feature or catecholaminergic polymorphic ventricular tachycardia [CPVT]) features of ATS, the proportion of individuals with an identified pathogenic variant was 75% (15/20) in those with typical ATS, 71% (5/7) in those with the cardiac phenotype alone, 100% (2/2) in those with periodic paralysis, and 7% (2/28) in those with CPVT [Kimura et al 2012].
In a single large kindred with the KCNJ2 p.Arg67Trp pathogenic variant, periodic paralysis was observed only in men, cardiac symptoms only in women, and congenital anomalies in both [Andelfinger et al 2002]. However, this apparent sex-limited bias in clinical presentation has not been confirmed [Donaldson et al 2003, Davies et al 2005].
A large five-generation family was found to have a deletion in KCNJ2 (c.271_282del12;p.Ala91_Leu94del) and more severe cardiac manifestations, including 4/10 individuals receiving an implantable cardioverter-defibrillator and 5/10 reporting life-threatening ventricular arrhythmias in childhood [Fernlund et al 2013]. These individuals had characteristic mild dysmorphic features, but only one had periodic paralysis.
Two pathogenic variants in KCNJ2 associated with a reduction in Kir2.1 current but without a dominant-negative effect on normal channels (p.Asn318Ser and p.Trp322Cys) were described in three individuals with an isolated cardiac phenotype: with premature ventricular contractions, ventricular tachycardia, or ventricular fibrillation [Limberg et al 2013].
A recent study examined the predictors of arrhythmias in 11 unrelated families with ATS (11 probands who were all female, 14 unaffected heterozygous family members with the KCNJ2 pathogenic variant, and 19 family members without the pathogenic variant) and found that a higher risk for arrhythmia, syncope, and/or cardiac arrest was associated with the presence of micrognathia, periodic paralysis, and prolonged duration of the descending portion of T-wave (Tpeak – Tend time) . It was also noted that the ATS clinical manifestations appeared to be more severe in females. The presence of a benign / likely benign variant in KCNH2 appeared to be associated with a higher risk for syncope [Krych et al 2017].
Penetrance
Non-penetrance is evident in 6%-20% of individuals with an identifiable pathogenic variant [Andelfinger et al 2002, Tristani-Firouzi et al 2002, Donaldson et al 2003].
Nomenclature
Although listed in OMIM, the following names for ATS are no longer in clinical use:
- Periodic paralysis, potassium-sensitive cardiodysrhythmic type
- Andersen cardiodysrhythmic periodic paralysis
Prevalence
The estimated prevalence of ATS is 0.08 – 0.1 per 100,000 [Horga et al 2013, Stunnenberg et al 2018].
Differential Diagnosis
Andersen-Tawil syndrome (ATS) should be considered in any individual presenting with periodic paralysis and ventricular arrhythmias or prominent U wave or prolonged QTc. Individuals with either episodic weakness or cardiac symptoms require careful evaluation by a neurologist and/or cardiologist as well as measurement of serum potassium concentration (baseline and during attacks of flaccid paralysis), a 12-lead ECG, a 24-hour Holter monitor, and possibly the long exercise protocol. The differential diagnosis depends on the initial presentation and includes the primary and secondary periodic paralyses, thyrotoxic periodic paralysis, and conditions associated with long QT.
Episodes of Flaccid Paralysis
Hypokalemic periodic paralysis is the most common periodic paralysis. Affected individuals may experience paralytic episodes with concomitant hypokalemia (<2.5 mmol/L), and occasionally may develop late-onset proximal myopathy. The paralytic attacks are characterized by reversible flaccid paralysis usually leading to paraparesis or tetraparesis but typically sparing the respiratory muscles and heart. The onset, duration, and severity of attacks, with the associated triggers, are similar to those in individuals with ATS. Weakness is improved with oral potassium ingestion. The cardiac and dysmorphic features of ATS are, however, absent in individuals with hypokalemic periodic paralysis. Molecular testing identifies pathogenic variants in CACNA1S or SCN4A in approximately 80% of affected individuals after secondary causes such as thyrotoxicosis, diuretic use, and renal (e.g., hyperaldosteronism, distal tubular acidosis) or gastrointestinal (e.g., vomiting, diarrheal illness) causes have been ruled out. Inheritance is autosomal dominant.
Hyperkalemic periodic paralysis is characterized by episodes of flaccid paralysis associated with normal or elevated ictal serum potassium concentrations (>5.0 mmol/U) and aggravated by potassium ingestion. Onset is in the first decade; episodes are briefer than those that occur in individuals with hypokalemic periodic paralysis. Electrical myotonia is evident in 50% of affected individuals. The cardiac and dysmorphic features of ATS are absent. Molecular testing reveals pathogenic variants in SCN4A in approximately 80% of individuals. Inheritance is autosomal dominant. Secondary forms of hyperkalemic periodic paralysis to rule out include adrenal insufficiency, hypoaldosteronism, and adverse effects of certain medications (e.g., ACE inhibitors, spironolactone, nonsteroidal anti-inflammatory drugs).
Thyrotoxic periodic paralysis (OMIM 188580, 613239, 614834) is a consideration in any individual with severe weakness and marked hypokalemia. Men, particularly Asians, are affected in greater numbers; however, thyrotoxic periodic paralysis may be seen in individuals of all ethnicities. Diagnosis is established by measurement of serum thyroid-stimulating hormone (TSH), T4, and T3 concentrations. In one series a pathogenic variant in an inwardly rectifying potassium (Kir) channel (encoded by KCNJ18) was identified in approximately one third of affected individuals [Ryan et al 2010].
Long QT Syndromes
See Long QT Syndrome, a review of similar phenotypes that are genetically diverse.
Palpitations, Syncope, or Cardiac Arrest
Syncopal episodes are often interpreted as a neurologic problem rather than arrhythmia. Physical examination and ECG should be part of the evaluation of syncope. Bidirectional ventricular tachycardia demonstrated on ECG may be seen with ATS, digitalis toxicity, and catecholaminergic polymorphic ventricular tachycardia (CPVT). A normal resting ECG with exercise-induced polymorphic arrhythmias is a clue to CPVT. Pathogenic variants in RYR2 or CASQ2 are causative. Inheritance is autosomal dominant [Tristani-Firouzi & Etheridge 2010].
Management
Evaluations Following Initial Diagnosis
To establish the extent of disease and needs in an individual diagnosed with Andersen-Tawil syndrome (ATS), the evaluations summarized in Table 2 (if not performed as part of the evaluation that led to the diagnosis) are recommended:
Table 2.
Recommended Evaluations Following Initial Diagnosis in Individuals with ATS
| Organ System | Evaluation | Comment |
|---|---|---|
| Cardiovascular | Baseline assessment | Performed by cardiologist familiar w/LQT management |
| 12-lead ECG & 24-hour Holter monitor | ||
| Serum potassium concentrations | Performed at baseline & during attacks of weakness | |
| Neurologic | Baseline assessment | Performed by neurologist familiar w/periodic paralysis |
| Electrophysiologic studies including long exercise protocol | ||
| Dental | Baseline assessment for dental abnormalities associated w/ATS | Follow up as needed |
| Musculoskeletal | Baseline assessment & to establish care w/orthopedist / spine surgeon if scoliosis identified | Follow up as needed |
| Miscellaneous/ Other | Serum TSH concentration | Verification that serum TSH concentration is w/in normal limits |
| Consultation w/clinical geneticist &/or genetic counselor |
LQT = long QT
TSH = thyroid-stimulating hormone
Treatment of Manifestations
Management of individuals with ATS requires the coordinated input of a neurologist familiar with the treatment of periodic paralysis and a cardiologist familiar with the treatment of cardiac arrhythmias. To date, no randomized clinical therapeutic trials have been conducted on ATS.
Management of attacks of episodic weakness depends on the associated serum potassium concentration:
- If the serum potassium concentration is low (<3.0 mmol/L), administer oral potassium (20-30 mEq/L) every 15-30 minutes (not to exceed 200 mEq in a 12-hour period) until the serum concentration normalizes often shortens the attack. As dysphagia is almost never a problem during attacks of paralysis, oral potassium replacement is the safest route. If intravenous potassium replacement is needed, a 5% mannitol solution instead of a saline or glucose solution (both of which may exacerbate weakness) is recommended. Close monitoring of serum potassium concentrations and ECG is necessary during potassium replacement therapy in an emergency setting to avoid secondary hyperkalemia.
- Whether a relative drop in serum potassium within the normal range causes episodic paralysis is not clear. If such cases are suspected, affected individuals can work with their physician to devise an individual potassium replacement regimen, with a goal of maintaining serum potassium levels in the high range of normal.
- Attacks of weakness when serum potassium concentration is high usually resolve within 60 minutes. Episodes may be shortened by ingesting carbohydrates or continuing mild exercise. Intravenous calcium gluconate is rarely necessary for management in an individual seen in an emergency setting.
Vasovagal syncope in individuals with ATS mandates a careful cardiology assessment [Airey et al 2009].
Prevention of Primary Manifestations
Prophylactic treatment aimed at reduction of attack frequency and severity can be achieved, as in other forms of periodic paralysis, with the following:
- Lifestyle and dietary modification to avoid known triggers
- Use of carbonic anhydrase inhibitors (acetazolamide: adults 125-1,000 mg daily and children 5-10 mg/kg/day divided 1-2x/day or dichlorphenamide 50-200 mg/1-2x/day). Use of potassium-sparing diuretic should be individualized based on patient needs.
- Daily use of slow-release potassium supplements, which may also be helpful in controlling attack rates in individuals prone to hypokalemia. Elevating the serum potassium concentration (>4 mEq/L) has the added benefit of narrowing the QT interval, thus reducing the risk of LQT-associated arrhythmias.
- An implantable cardioverter-defibrillator in individuals with tachycardia-induced syncope [Chun et al 2004]
- Empiric treatment with flecainide [Bökenkamp et al 2007, Fox et al 2008, Pellizzón et al 2008] should be considered for significant, frequent ventricular arrhythmias in the setting of reduced left ventricular function [Tristani-Firouzi & Etheridge 2010]. A prospective open label study in ten individuals with ATS and a confirmed KCNJ2 pathogenic variant tested the effect of flecainide, a type 1c antiarrhythmic, for the prevention of cardiac arrhythmias [Miyamoto et al 2015]. Outcomes included a 24-hour Holter monitor before and after treatment and a treadmill exercise test. Flecainide was found to significantly reduce the number of ventricular arrhythmias seen on Holter monitor and to suppress exercise-induced ventricular arrhythmias. Individuals were then followed for a mean of 23 months and no syncope or cardiac arrest was documented. Other case studies have reported beneficial effects with flecainide [Statland et al 2018]. A recent study showed that fleicainide suppresses arrhythmogenicity through Na+/Ca2+ exchanger flux in induced pluripotent stem cells derived from patients with ATS [Kuroda et al 2017]. Thus, flecainide may reduce cardiac arrhythmias in ATS.
Prevention of Secondary Complications
Cardiologists should be aware that some antiarrhythmic drugs (e.g., lidocaine, mexiletine, propafenone, quinidine), particularly Class I drugs, may paradoxically exacerbate the neuromuscular symptoms and should be used cautiously in individuals with ATS.
Although malignant hyperthermia has not been reported in ATS, appropriate anesthetic precautions should be undertaken, as with individuals with other forms of periodic paralysis.
Surveillance
For asymptomatic individuals with a KCNJ2 pathogenic variant, annual screening including a 12-lead ECG and 24-hour Holter monitoring is desirable, followed by referral to a cardiologist if abnormalities are identified.
Agents/Circumstances to Avoid
Affected individuals should avoid medications known to prolong QT intervals. See CredibleMeds® for a complete and updated list.
Salbutamol inhalers, which may be used in the treatment of primary hyperkalemic periodic paralysis, should be avoided because of the potential for exacerbation of cardiac arrhythmias.
Thiazide and other potassium-wasting diuretics may provoke drug-induced hypokalemia and could aggravate the QT interval prolongation.
Evaluation of Relatives at Risk
It is appropriate to evaluate relatives at risk in order to identify as early as possible those who would benefit from initiation of treatment and preventive measures:
- If the pathogenic variant in the family is known, molecular genetic testing can be used to clarify the genetic status of at-risk relatives.
- If the pathogenic variant in the family is not known, detailed neurologic and cardiologic evaluation, 12-lead ECG, and 24-hour Holter monitoring can be used to clarify the disease status of at-risk relatives.
See Genetic Counseling for issues related to testing of at-risk relatives for genetic counseling purposes.
Pregnancy Management
The rarity of ATS and the paucity of reports pertaining to pregnancy in women with ATS make an evidence-based approach to pregnancy management difficult to formulate. One case study reported an uneventful pregnancy, with increased episodes of weakness but reduced ventricular ectopy compared to the pre-pregnancy period [Subbiah et al 2008]. However, as data are limited, a multidisciplinary approach to individual care and anticipation of increased risk (as can be seen in those with long QT syndrome) seems reasonable.
Therapies Under Investigation
Search ClinicalTrials.gov in the US and www.ClinicalTrialsRegister.eu in Europe for information on clinical studies for a wide range of diseases and conditions. Note: There may not be clinical trials for this disorder.