-
Opioid Use Disorder
Wikipedia
Opioid use disorder Other names Opioid addiction, [1] problematic opioid use, [1] opioid abuse, [2] opioid dependence [3] Molecular structure of morphine Specialty Addiction Medicine , Psychiatry Symptoms Strong desire to use opioids, increased tolerance to opioids, failure to meet obligations, trouble with reducing use, withdrawal syndrome with discontinuation [4] [5] Complications Opioid overdose , hepatitis C , marriage problems, unemployment, poverty [4] [5] Duration Long term [6] Causes Opioids [3] Diagnostic method Based on criteria in the DSM-5 [4] Differential diagnosis Alcoholism Treatment Opioid replacement therapy , behavioral therapy , twelve-step programs , take home naloxone [7] [8] [9] Medication Buprenorphine , methadone , naltrexone [7] [10] Frequency 27 million (c. 0.4%) [11] [4] Deaths 122,000 (2015) [12] Opioid use disorder ( OUD ) is a substance use disorder relating to the use of an opioid . Any such disorder causes significant impairment or distress. [3] Signs of the disorder include a strong desire to use opioids, increased tolerance to opioids, difficulty fulfilling obligations, trouble reducing use, and withdrawal symptoms with discontinuation. [4] [5] Opioid withdrawal symptoms may include nausea, muscle aches, diarrhea, trouble sleeping, agitation, and a low mood. [5] Addiction and dependence are components of a substance use disorder. [13] Complications may include opioid overdose , suicide , HIV/AIDS , hepatitis C , and problems at school, work, or home. [4] [5] Opioids include substances such as heroin , morphine , fentanyl , codeine , dihydrocodeine , oxycodone , and hydrocodone . [5] [6] In the United States, a majority of heroin users begin by using prescription opioids, that may also be bought illegally. [14] [15] Risk factors for misuse include a history of substance use, substance use among family and friends, mental illness, low socioeconomic status, and race. [16] [17] Diagnosis may be based on criteria by the American Psychiatric Association in the DSM-5 . [4] If more than two of eleven criteria are present during a year, the diagnosis is said to be present. [4] If a person is appropriately taking opioids for a medical condition, issues of tolerance and withdrawal do not apply. [4] Individuals with an opioid use disorder are often treated with opioid replacement therapy using methadone or buprenorphine . [7] Being on such treatment reduces the risk of death. [7] Additionally, individuals may benefit from cognitive behavioral therapy , other forms of support from mental health professionals such as individual or group therapy, twelve-step programs , and other peer support programs. [8] The medication naltrexone may also be useful to prevent relapse. [10] Naloxone is useful for treating an opioid overdose and giving those at risk naloxone to take home is beneficial. [9] In 2013, opioid use disorders affected about 0.4% of people. [4] As of 2016, about 27 million people are affected. [11] Long term opioid use occurs in about 4% of people following their use for trauma or surgery related pain. [18] Onset is often in young adulthood. [4] Males are affected more often than females. [4] It resulted in 122,000 deaths worldwide in 2015, [12] up from 18,000 deaths in 1990. [19] In the United States during 2016, there were more than 42,000 deaths due to opioid overdose, of which more than 15,000 were the result of heroin use. [20] Contents 1 Signs and symptoms 1.1 Withdrawal 1.2 Opioid intoxication 1.3 Opioid overdose 2 Cause 3 Mechanism 3.1 Addiction 3.2 Dependence 3.3 Opioid receptors 3.3.1 118A>G variant 3.3.2 Non-opioid receptor genes 4 Diagnosis 5 Prevention 5.1 Opioid related deaths 6 Management 6.1 Medications 6.1.1 Methadone 6.1.2 Buprenorphine 6.1.3 Other opioids 6.1.4 Naltrexone 6.2 Behavioral therapy 6.2.1 Cognitive behavioral therapy 6.2.2 Twelve-step programs 6.2.3 Digital care programs 7 Epidemiology 7.1 United States 8 History 9 See also 10 References 11 External links Signs and symptoms [ edit ] Material used for intravenous injection of opioids Signs and symptoms include: [4] [5] Drug seeking behavior Increased use over time Legal or social ramifications secondary to drug use Multiple prescriptions from different providers Multiple medical complications from drug use ( HIV/AIDS , hospitalizations, abscesses ) Opioid cravings Withdrawal symptoms Addiction and dependence are components of a substance use disorder and addiction represents the more severe form. [13] Opioid dependence can occur as physical dependence , psychological dependence , or both. [21] Withdrawal [ edit ] Opioid withdrawal can occur with a sudden decrease in, or the cessation of opioids after prolonged use. [22] [23] Onset of withdrawal depends on which opioid was used last. [24] With heroin this typically occurs five hours after use, while with methadone it might not occur until two days later. [24] The length of time that major symptoms occur also depends on the opioid used. [24] For heroin withdrawal, symptoms are typically greatest at two to four days, and can last for up to two weeks. [25] [24] Less significant symptoms may remain for an even longer period, in which case the withdrawal is known as post-acute-withdrawal syndrome . [24] Agitation [4] Anxiety [4] Muscle pains [4] Increased tearing [4] Trouble sleeping [4] Runny nose [4] Sweating [4] Yawning [4] Goose bumps [4] Dilated pupils [4] Diarrhea [4] Fast heart rate [24] High blood pressure [24] Abdominal cramps [24] Shakiness [24] Cravings [24] Sneezing [24] Treatment of withdrawal may include methadone and buprenorphine. ... However, the exact mechanisms involved are unclear, leading to debate regarding where the influence of biology and free will. [33] [34] Mechanism [ edit ] Addiction [ edit ] Addiction is a brain disorder characterized by compulsive drug use despite adverse consequences. [13] [35] [36] [37] Addiction is a component of a substance use disorder and represents the most severe form of the disorder. [13] Overexpression of the gene transcription factor ΔFosB in the nucleus accumbens plays a crucial role in the development of an addiction to opioids and other addictive drugs by sensitizing drug reward and amplifying compulsive drug-seeking behavior. [35] [38] [39] [40] Like other addictive drugs , overuse of opioids leads to increased ΔFosB expression in the nucleus accumbens . [38] [39] [40] [41] Opioids affect dopamine neurotransmission in the nucleus accumbens via the disinhibition of dopaminergic pathways as a result of inhibiting the GABA -based projections to the ventral tegmental area (VTA) from the rostromedial tegmental nucleus (RMTg), which negatively modulate dopamine neurotransmission. [42] [43] In other words, opioids inhibit the projections from the RMTg to the VTA, which in turn disinhibits the dopaminergic pathways that project from the VTA to the nucleus accumbens and elsewhere in the brain. [42] [43] Neuroimaging has shown functional and structural alterations in the brain. [44] A 2017 study showed that chronic intake of opioids, such as heroin, may cause long-term effects in the orbitofrontal area (OFC), which is essential for regulating reward-related behaviors, emotional responses, and anxiety. [45] Moreover, neuroimaging and neuropsychological studies demonstrated dysregulation of circuits associated with emotion, stress and high impulsivity. [46] Dependence [ edit ] Drug dependence is an adaptive state associated with a withdrawal syndrome upon cessation of repeated exposure to a stimulus (e.g., drug intake). [35] [36] [37] Dependence is a component of a substance use disorder . [13] [47] Opioid dependence can manifest as physical dependence , psychological dependence , or both. [21] [36] [47] Increased brain-derived neurotrophic factor (BDNF) signaling in the ventral tegmental area (VTA) has been shown to mediate opioid-induced withdrawal symptoms via downregulation of insulin receptor substrate 2 (IRS2), protein kinase B (AKT), and mechanistic target of rapamycin complex 2 (mTORC2). [35] [48] As a result of downregulated signaling through these proteins, opiates cause VTA neuronal hyperexcitability and shrinkage (specifically, the size of the neuronal soma is reduced). [35] It has been shown that when an opiate-naive person begins using opiates in concentrations that induce euphoria , BDNF signaling increases in the VTA. [49] Upregulation of the cyclic adenosine monophosphate (cAMP) signal transduction pathway by cAMP response element binding protein (CREB), a gene transcription factor , in the nucleus accumbens is a common mechanism of psychological dependence among several classes of drugs of abuse. [21] [35] Upregulation of the same pathway in the locus coeruleus is also a mechanism responsible for certain aspects of opioid-induced physical dependence . [21] [35] Opioid receptors [ edit ] A genetic basis for the efficacy of opioids in the treatment of pain has been demonstrated for several specific variations; however, the evidence for clinical differences in opioid effects is ambiguous. ... If initial doses during the beginning of treatment are too high or are concurrent with illicit opioid use, this may present an increased risk of death from overdose. [7] In addition, enrollment in methadone maintenance has the potential to reduce the transmission of infectious diseases associated with opiate injection, such as hepatitis and HIV. [81] The principal effects of methadone maintenance are to relieve narcotic craving, suppress the abstinence syndrome, and block the euphoric effects associated with opiates. ... LAAM produced long-lasting effects, which allowed the person receiving treatment to visit a clinic only three times per week, as opposed to daily as with methadone. [126] In 2001, levacetylmethadol was removed from the European market due to reports of life-threatening ventricular rhythm disorders . [127] In 2003, Roxane Laboratories, Inc. discontinued Orlaam in the US. [128] See also [ edit ] Benzodiazepine withdrawal syndrome Doctor shopping Hyperkatifeia , hypersensitivity to emotional distress in the context of opioid abuse Physical dependence Post-acute-withdrawal syndrome Prescription drug abuse References [ edit ] ^ a b "FDA approves first buprenorphine implant for treatment of opioid dependence" .
-
Gluten-Related Disorders
Wikipedia
The effectiveness of the treatment depends on the elapsed time from the onset of the ataxia until diagnosis, because the death of neurons in the cerebellum as a result of gluten exposure is irreversible. [29] [30] Gluten ataxia accounts for 40% of ataxias of unknown origin and 15% of all ataxias. [29] [31] Less than 10% of people with gluten ataxia present any gastrointestinal symptom, yet about 40% have intestinal damage. [29] Non-celiac gluten sensitivity (NCGS) [ edit ] Main article: Non-celiac gluten sensitivity Non-celiac gluten sensitivity (NCGS), or gluten sensitivity (GS), [1] is a possible syndrome in which people develop a variety of intestinal and/or extraintestinal symptoms that improve when gluten is removed from the diet, [32] after coeliac disease and wheat allergy are excluded. [33] NCGS, which is possibly immune-mediated, now appears to be more common than coeliac disease, [34] with a prevalence estimated to be 6–10 times higher. [35] Gastrointestinal symptoms, which resemble those of irritable bowel syndrome (IBS), [32] [36] may include any of the following: abdominal pain , bloating , bowel habit abnormalities (either diarrhea or constipation ), [36] [37] nausea , aerophagia , gastroesophageal reflux disease , and aphthous stomatitis . [33] [36] Extra-intestinal symptoms, which can be the only manifestation of NCGS even in absence of gastrointestinal symptoms, may be any of the following: headache or migraine , “foggy mind”, fatigue , [33] [36] [37] fibromyalgia , [37] [38] [39] joint and muscle pain, [33] [36] [37] leg or arm numbness , [33] [36] [37] tingling of the extremities, [33] [36] dermatitis ( eczema or skin rash ), [33] [36] atopic disorders , [33] allergy to one or more inhalants, foods or metals [33] [37] (such as mites , graminaceae , parietaria , cat or dog hair, shellfish , or nickel ), [37] depression , [33] [36] [37] anxiety , [37] anemia , [33] [36] iron-deficiency anemia , folate deficiency , asthma , rhinitis , eating disorders , [37] or autoimmune diseases . [33] Among extra-intestinal manifestations, NCGS seems to be involved in some neuropsychiatric disorders , [40] principally schizophrenia , [12] [36] autism [12] [36] [37] and peripheral neuropathy , [12] [36] and also ataxia [12] and attention deficit hyperactivity disorder (ADHD). [33] Gluten is likely responsible for the appearance of symptoms, but it has been suggested than in a subgroup of people with NCGS and symptoms like IBS, other components of wheat and related grains (oligosaccharides like fructans), or other plant proteins contained in gluten-containing cereals (agglutinins, lectins, and amylase trypsin inhibitors (ATIs)) may play a role in the development of gastrointestinal symptoms. [17] ATIs are about 2–4% of the total protein in modern wheat and 80–90% in gluten. [33] In a review of May 2015 published in Gastroenterology , Fasano et al. conclude that ATIs may be the inducers of innate immunity in people with coeliac disease or NCGS. [33] As of 2019, reviews conclude that although FODMAPs present in wheat and related grains may play a role in non-celiac gluten sensitivity, they only explain certain gastrointestinal symptoms, such as bloating , but not the extra-digestive symptoms that people with non-celiac gluten sensitivity may develop, such as neurological disorders , fibromyalgia , psychological disturbances, and dermatitis . [41] [42] [33] As occurs in people with coeliac disease, the treatment is a gluten-free diet (GFD) strict and maintained, without making any dietary transgression. [37] Whereas coeliac disease requires adherence to a strict lifelong gluten-free diet, it is not yet known whether NCGS is a permanent, or a transient condition. [22] [37] The results of a 2017 study suggest that NCGS may be a chronic disorder, as is the case with celiac disease. [42] Theoretically, a trial of gluten reintroduction to observe reaction after 1–2 years of strict gluten-free diet might be advisable. [37] Approximately one third of personas with NCGS continue having symptoms despite gluten withdrawal. ... Of people with pseudo-exfoliation syndrome , 25% showed increased levels of anti-gliadin IgA. [49] Other people that are also at risk are those taking gluten despite having the disorder, or whose family members have CD. ... Therefore gluten intake should be limited before or even after the diagnosis. [50] One fourth of people with Sjögren's syndrome had responses to gluten, of 5 that had positive response to gluten, only one could be confirmed as CD and another was potentially GSE [ clarification needed ] , the remaining 3 appear to be gluten-sensitive. ... The symbol, which is protected as a trademark in Europe and the United States and is covered by worldwide copyright, can be represented in any colour. [81] [82] Research [ edit ] Research has attempted to discern, by double-blind placebo-controlled trials , between a "fad component" to the recent popularity of the gluten-free diet and an actual sensitivity to gluten or other components of wheat. [33] [36] [83] In a 2013 double-blind , placebo-controlled challenge (DBPC) by Biesiekierski et al. in a few people with irritable bowel syndrome , the authors found no difference between gluten or placebo groups and the concept of NCGS as a syndrome was questioned. ... "Gluten sensitivity in patients with primary Sjögren's syndrome". Scand. J. Gastroenterol. (Research Support, Non-U.S.
-
Extinction (Neurology)
Wikipedia
Recent literature suggests that unilateral extinction patients not only fail to respond to the contralateral external space, but also to internally represented stimuli with patients frequently locating the details of the left side on the right side. [3] Research and characteristics of extinction [ edit ] German neurologists documented clinical descriptions of extinction a century ago, but the syndrome subsequently received less systematic attention than other classical neurological syndromes in part due to the rareness of suitable theoretical ideas. ... There is also much excitement about the possibility of relating awareness to neural substrates in extinction studies. [3] In addition to revealing the critical lesion sites associated with the various clinical manifestations of visual neglect, a key message of the current investigation is that there is a need to develop more sensitive and nuanced assessment tools to characterize the different facets of this heterogeneous syndrome. It will be important to bring laboratory tests into the clinic in an effort to identify specific cognitive functions by examining each in isolation thus combining more specific descriptions extinction with better clinical measures that isolate specific cognitive functions to yield more consistent lesion mapping results in the future. [4] Grouping effect in extinction [ edit ] Figure from a paper by J. ... It is noteworthy that the right Temporoparietal Junction (TPJ) has been linked to a number of cognitive functions that suggest a role in modulating competitive interactions between stimulus representations, which would converge with the importance of this area for the attentional deficit displayed by extinction patients. [5] The critical lesion site responsible for the syndrome has been debated for more than a decade.
-
Substance Dependence
Wikipedia
Within the framework of the 4th edition of the Diagnostic and Statistical Manual of Mental Disorders ( DSM-IV ), substance dependence is redefined as a drug addiction, and can be diagnosed without the occurrence of a withdrawal syndrome. [6] It was described accordingly: "When an individual persists in use of alcohol or other drugs despite problems related to use of the substance, substance dependence may be diagnosed. ... Psychological stress may also result if the substance is not re-introduced. [13] Infants also suffer from substance withdrawal, known as neonatal abstinence syndrome (NAS), which can have severe and life-threatening effects. ... It is ironic and unfortunate that the Manual avoids use of the term addiction, which provides the best description of the clinical syndrome. ^ a b "Substance use disorder" . ... Retrieved 21 December 2020 . ^ "Supporting mothers with opioid addiction is the best bet in fighting neonatal abstinence syndrome" . sheknows.com . 10 May 2017. ... Retrieved 26 February 2015 . - pdf v t e Psychoactive substance-related disorder General SID Substance intoxication / Drug overdose Substance-induced psychosis Withdrawal : Craving Neonatal withdrawal Post-acute-withdrawal syndrome (PAWS) SUD Substance abuse / Substance-related disorders Physical dependence / Psychological dependence / Substance dependence Combined substance use SUD Polysubstance dependence SID Combined drug intoxication (CDI) Alcohol SID Cardiovascular diseases Alcoholic cardiomyopathy Alcohol flush reaction (AFR) Gastrointestinal diseases Alcoholic liver disease (ALD): Alcoholic hepatitis Auto-brewery syndrome (ABS) Endocrine diseases Alcoholic ketoacidosis (AKA) Nervous system diseases Alcohol-related dementia (ARD) Alcohol intoxication Hangover Neurological disorders Alcoholic hallucinosis Alcoholic polyneuropathy Alcohol-related brain damage Alcohol withdrawal syndrome (AWS): Alcoholic hallucinosis Delirium tremens (DTs) Fetal alcohol spectrum disorder (FASD) Fetal alcohol syndrome (FAS) Korsakoff syndrome Positional alcohol nystagmus (PAN) Wernicke–Korsakoff syndrome (WKS, Korsakoff psychosi
-
Distal Radius Fracture
Wikipedia
Swelling and displacement can cause compression on the median nerve which results in acute carpal tunnel syndrome and requires prompt treatment. Very rarely, pressure on the muscle components of the hand or forearm is sufficient to create a compartment syndrome which can manifest as severe pain and sensory deficits in the hand. [5] Malreduced distal radius fracture demonstrating the deformity in the wrist "Dinner fork" deformity Complications [ edit ] Nonunion is rare; almost all of these fractures heal. ... This arthritis can worsen over time. [6] Displaced fractures of the ulnar styloid base associated with a distal radius fracture result in instability of the DRUJ and resulting loss of forearm rotation. [ citation needed ] Nerve injury, especially of the median nerve and presenting as carpal tunnel syndrome, is commonly reported following distal radius fractures. ... This can be due to the tendon coming in contact with protruding bone or with hardware placed following surgical procedures. [ citation needed ] Complex regional pain syndrome is also associated with distal radius fractures, and can present with pain, swelling, changes in color and temperature, and/or joint contracture.
-
Ataxia-Telangiectasia
Omim
Neurologic Manifestations AT may be the most common syndromic progressive cerebellar ataxia of early childhood. ... See 208910 and 208920 for AT-like syndromes. Rare cases of AT patients with milder manifestations of the clinical or cellular characteristics of the disease have been reported and have been designated 'AT variants.' ... Jaspers et al. (1988) reported the results of complementation studies on fibroblast strains from 50 patients with AT or Nijmegen breakage syndrome (NBS; 251260), using the radioresistant DNA replication characteristic as a marker. ... Curry et al. (1989) used the designation AT(Fresno) (607585.0014) for the V1 disorder in twin girls who had clinical features combining those of ataxia-telangiectasia and the Nijmegen breakage syndrome. Complementation studies with Sendai virus-mediated fusion of fibroblast cell lines showed complementation with AT groups A, C, and E but not with the cell line from a patient with the Nijmegen breakage syndrome. ... However, Komatsu et al. (1996) could demonstrate that the gene for the V2 form of Nijmegen breakage syndrome is not located on chromosome 11.ATM, C11orf65, NLRP2, H2AX, AFP, APTX, ATR, NBN, CHEK1, TP53, MRE11, TRIM29, SETX, CDKN1A, FXN, CHEK2, TCL1B, TRBV20OR9-2, TCL1A, BRCA1, CD38, THY1, ETV6, MDM2, FEV, RAD50, BRCA2, BRAF, PRKDC, XRS, NPAT, SERPINA1, PIK3CD, PIK3CB, SLC12A9, ACAT1, FANCD2, PIK3CA, IRS2, GADD45A, PIK3CG, ISG15, AGT, STK11, BLM, TSHR, IGH, MYC, KRAS, KAT5, ERBB2, JAK3, CYSLTR1, IRS1, CXCL8, IL4, PNKP, GYPA, MMP9, HPRT1, DYNC2H1, FN1, PPM1D, MTCP1, TERF1, H1-0, GH1, GRIA4, HLA-DRB1, LIG4, CDK1, MIR421, RNF168, CCL24, CAT, CCNB1, TPO, MIR30A, ARSA, COL26A1, CD40, CD40LG, FAS, SLC1A5, DHFR, ACTB, PTCH1, TNF, TG, CUL5, TFRC, BMS1, PDLIM7, VEGFA, NCOR2, UVRAG, U2AF1, HERPUD1, TYR, TOP1, TYMS, TGFB1, TGM2, TM7SF2, TP53BP1, VHL, TOP2A, MDC1, UBE3C, KIAA0753, XPA, EXO1, CCNB2, H4C11, H4C3, H4C8, H4C2, H4C6, H4C4, H4C1, H4C9, SMC1A, FOSL1, H4C5, H4C13, CCDC6, H4C14, ARHGEF5, ZNF148, TP63, YY1, TNFSF10, HESX1, XRCC5, XRCC1, APLN, H4C12, TIMELESS, ACACA, FGF21, RANBP9, SPSB1, CENPV, SLC30A8, FSIP1, CNKSR3, PLB1, OR2AG1, HT, DCBLD2, RBM45, CANT1, H4-16, DTD1, SLFN11, PARP10, ATRIP, SESN2, SLC38A1, FLCN, TMEM196, ZC3H12D, MIR708, LOC110806263, LOC102724971, LOC102724334, LOC102723407, PGR-AS1, COMMD3-BMI1, KLRC4-KLRK1, POTEF, MALAT1, MUC5B, H4C15, MIR449A, PLF, MIR203A, IYD, C12orf75, ULBP2, NHEJ1, PSME3, TTC21B, SETD2, DLL1, IGHV3-69-1, IGHV3OR16-7, INTU, HAVCR1, IFT172, HINFP, SUMF2, IL17RA, KLRK1, TREX1, PTGDR2, FSTL1, STMN2, CKAP4, GLYAT, CD274, GPR132, IL22, IFT80, AAGAB, NABP2, SMAP2, XYLT2, CLSPN, ACE2, WDR19, WDR35, TRAT1, ENAH, RNPC3, SLC30A10, LRRC59, NEIL3, WRAP53, H2BS1, TERT, UPF1, TERC, CRYGD, EZH2, ETS1, ELANE, EGFR, EGF, EDN1, E2F3, E2F1, DRD2, DPYD, DNMT1, DLG4, DECR1, DCK, CYP2D6, CTLA4, CSN2, F9, FABP1, FANCA, GABPA, HLA-DPA1, HIF1A, GYPE, GYPB, GTF2H4, GTF2H3, GSTP1, GAA, FANCC, G6PD, FSHR, FOS, FLT3, FOXO3, FCGRT, FCER1G, CSF1R, CRYAB, TRG, CES1, BAX, ATD, SERPINC1, AR, APOE, APOC3, BIRC3, BIRC2, ANGPT2, ANGPT1, ALOX5, AKT2, AKT1, AGTR2, AGTR1, ADRB2, PARP1, BCL2, BIK, BMI1, CAV1, CDKN2A, CDK2, CDC25C, CD69, CD59, CD5L, CCNH, CASP9, BMP1, CASP8, CASP3, CASP1, VPS51, BUB1B, BUB1, BTF3, HLA-DPB1, HLA-DQB1, HLA-DRB3, HOXD13, RPE65, RGS2, RET, RASGRF1, RAG2, RAG1, RAD51, RAB1A, PTGS1, TAS2R38, PSMC1, PSMB9, PSMB6, RELN, PRSS1, PPARG, PML, RPS6KA3, SGCA, SLC2A1, SSB, TRB, TRA, TBXAS1, TBXA2R, SYP, STAT5B, STAT3, SPTBN2, SLC5A5, SOD1, SOAT1, SNRPN, SNAP25, SLC22A2, SLC15A1, SLC6A12, PLK1, PLAT, PLA2G4A, IL1B, LOX, LIG3, LIG1, LBR, KPNA4, ITGA2B, IL2RG, IL1A, MARS1, IGF1, IFNB1, IFNA13, IFNA1, HSPA5, HSPA4, HSPA1B, LTC4S, MAS1, ENPP2, MUC5AC, PAH, NPPA, NME1, NFKBIA, NFE2L2, NFATC1, NCAM1, MUC4, MGMT, MUC1, MTHFR, ATP6, MSH2, MMP3, MMP2, ATXN3, H3P10
-
Pulmonary Contusion
Wikipedia
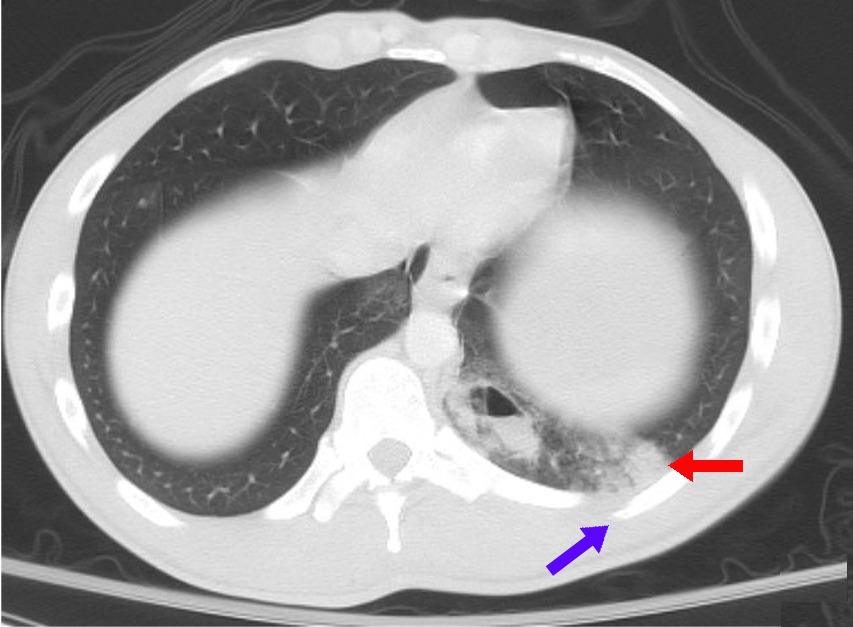
Pulmonary contusion is associated with complications including pneumonia and acute respiratory distress syndrome , and it can cause long-term respiratory disability. ... Macrophages , neutrophils , and other inflammatory cells and blood components can enter the lung tissue and release factors that lead to inflammation, increasing the likelihood of respiratory failure. [37] In response to inflammation, excess mucus is produced, potentially plugging parts of the lung and leading to their collapse. [24] Even when only one side of the chest is injured, inflammation may also affect the other lung. [37] Uninjured lung tissue may develop edema, thickening of the septa of the alveoli, and other changes. [38] If this inflammation is severe enough, it can lead to dysfunction of the lungs like that seen in acute respiratory distress syndrome. [39] Ventilation/perfusion mismatch [ edit ] Normally, the ratio of ventilation to perfusion is about one-to-one; the volume of air entering the alveoli ( ventilation ) is about equal to that of blood in the capillaries around them ( perfusion ). [40] This ratio is reduced in pulmonary contusion; fluid-filled alveoli cannot fill with air, oxygen does not fully saturate the hemoglobin , and the blood leaves the lung without being fully oxygenated. [41] Insufficient inflation of the lungs, which can result from inadequate mechanical ventilation or an associated injury such as flail chest, can also contribute to the ventilation/perfusion mismatch. [31] As the mismatch between ventilation and perfusion grows, blood oxygen saturation is reduced. [41] Pulmonary hypoxic vasoconstriction, in which blood vessels near the hypoxic alveoli constrict (narrow their diameter) in response to the lowered oxygen levels, can occur in pulmonary contusion. [27] The vascular resistance increases in the contused part of the lung, leading to a decrease in the amount of blood that flows into it, [38] directing blood to better-ventilated areas. [27] Although reducing blood flow to the unventilated alveoli is a way to compensate for the fact that blood passing unventilated alveoli is not oxygenated, [27] the oxygenation of the blood remains lower than normal. [40] If it is severe enough, the hypoxemia resulting from fluid in the alveoli cannot be corrected just by giving supplemental oxygen; this problem is the cause of a large portion of the fatalities that result from trauma. [41] Diagnosis [ edit ] To diagnose pulmonary contusion, health professionals use clues from a physical examination, information about the event that caused the injury, and radiography . [17] Laboratory findings may also be used; for example, arterial blood gasses may show insufficient oxygen and excessive carbon dioxide even in someone receiving supplemental oxygen. [35] However, blood gas levels may show no abnormality early in the course of pulmonary contusion. [23] X-ray [ edit ] A chest X-ray showing right sided (seen on the left of the picture) pulmonary contusion associated with rib fractures and subcutaneous emphysema Chest X-ray is the most common method used for diagnosis, [37] and may be used to confirm a diagnosis already made using clinical signs . [20] Consolidated areas appear white on an X-ray film. [42] Contusion is not typically restricted by the anatomical boundaries of the lobes or segments of the lung. [27] [43] [44] The X-ray appearance of pulmonary contusion is similar to that of aspiration , [32] and the presence of hemothorax or pneumothorax may obscure the contusion on a radiograph. [25] Signs of contusion that progress after 48 hours post-injury are likely to be actually due to aspiration, pneumonia, or ARDS. [10] Although chest radiography is an important part of the diagnosis, it is often not sensitive enough to detect the condition early after the injury. [35] In a third of cases, pulmonary contusion is not visible on the first chest radiograph performed. [7] It takes an average of six hours for the characteristic white regions to show up on a chest X-ray, and the contusion may not become apparent for 48 hours. [7] [27] [43] When a pulmonary contusion is apparent in an X-ray, it suggests that the trauma to the chest was severe and that a CT scan might reveal other injuries that were missed with X-ray. [2] Computed tomography [ edit ] A chest CT scan revealing pulmonary contusions, pneumothorax , and pseudocysts Computed tomography (CT scanning) is a more sensitive test for pulmonary contusion, [6] [33] and it can identify abdominal , chest, or other injuries that accompany the contusion. [38] In one study, chest X-ray detected pulmonary contusions in 16.3% of people with serious blunt trauma, while CT detected them in 31.2% of the same people. [45] Unlike X-ray, CT scanning can detect the contusion almost immediately after the injury. [43] However, in both X-ray and CT a contusion may become more visible over the first 24–48 hours after trauma as bleeding and edema into lung tissues progress. [46] CT scanning also helps determine the size of a contusion, which is useful in determining whether a patient needs mechanical ventilation; a larger volume of contused lung on CT scan is associated with an increased likelihood that ventilation will be needed. [43] CT scans also help differentiate between contusion and pulmonary hematoma , which may be difficult to tell apart otherwise. [47] However, pulmonary contusions that are visible on CT but not chest X-ray are usually not severe enough to affect outcome or treatment. [37] Ultrasound [ edit ] An ultrasound image showing early pulmonary contusion, at this moment not visible on radiography. ... Airbags in combination with seat belts can protect vehicle occupants by preventing the chest from striking the interior of the vehicle during a collision, and by distributing forces involved in the crash more evenly across the body. [6] However, in rare cases, an airbag causes pulmonary contusion in a person who is not properly positioned when it deploys. [50] Child restraints such as carseats protect children in vehicle collisions from pulmonary contusion. [51] Equipment exists for use in some sports to prevent chest and lung injury; for example, in softball the catcher is equipped with a chest protector. [52] Athletes who do not wear such equipment, such as basketball players, can be trained to protect their chests from impacts. [52] Protective garments can also prevent pulmonary contusion in explosions. [53] Although traditional body armor made from rigid plates or other heavy materials protects from projectiles generated by a blast, it does not protect against pulmonary contusion, because it does not prevent the blast's shock wave from being transferred to the lung. [53] Special body armor has been designed for military personnel at high risk for blast injuries; these garments can prevent a shock wave from being propagated across the chest wall to the lung, and thus protect wearers from blast lung injuries. [53] These garments alternate layers of materials with high and low acoustic impedance (the product of a material's density and a wave's velocity through it) in order to "decouple" the blast wave, preventing its propagation into the tissues. [53] Treatment [ edit ] No treatment is known to speed the healing of a pulmonary contusion; the main care is supportive. [39] Attempts are made to discover injuries accompanying the contusion, [20] to prevent additional injury, and to provide supportive care while waiting for the contusion to heal. [39] Monitoring, including keeping track of fluid balance , respiratory function, and oxygen saturation using pulse oximetry is also required as the patient's condition may progressively worsen. [54] Monitoring for complications such as pneumonia and acute respiratory distress syndrome is of critical importance. [55] Treatment aims to prevent respiratory failure and to ensure adequate blood oxygenation . [16] [23] Supplemental oxygen can be given and it may be warmed and humidified. [41] When the contusion does not respond to other treatments, extracorporeal membranous oxygenation may be used, pumping blood from the body into a machine that oxygenates it and removes carbon dioxide prior to pumping it back in. [56] Ventilation [ edit ] Mechanical ventilation may be required if pulmonary contusion causes inadequate oxygenation. ... Noninvasive positive pressure ventilation including continuous positive airway pressure (CPAP) and bi-level positive airway pressure (BiPAP), may be used to improve oxygenation and treat atelectasis: air is blown into the airways at a prescribed pressure via a face mask. [39] Noninvasive ventilation has advantages over invasive methods because it does not carry the risk of infection that intubation does, and it allows normal coughing, swallowing, and speech. [39] However, the technique may cause complications; it may force air into the stomach or cause aspiration of stomach contents, especially when level of consciousness is decreased. [4] People with signs of inadequate respiration or oxygenation may need to be intubated [7] and mechanically ventilated . [12] Mechanical ventilation aims to reduce pulmonary edema and increase oxygenation. [27] Ventilation can reopen collapsed alveoli, but it is harmful for them to be repeatedly opened, and positive pressure ventilation can also damage the lung by overinflating it. [57] Intubation is normally reserved for when respiratory problems occur, [7] but most significant contusions do require intubation, and it may be done early in anticipation of this need. [4] People with pulmonary contusion who are especially likely to need ventilation include those with prior severe lung disease or kidney problems; the elderly; those with a lowered level of consciousness; those with low blood oxygen or high carbon dioxide levels; and those who will undergo operations with anesthesia . [41] Larger contusions have been correlated with a need for ventilation for longer periods of time. [15] Pulmonary contusion or its complications such as acute respiratory distress syndrome may cause lungs to lose compliance (stiffen), so higher pressures may be needed to give normal amounts of air [4] and oxygenate the blood adequately. [33] Positive end-expiratory pressure (PEEP), which delivers air at a given pressure at the end of the expiratory cycle, can reduce edema and keep alveoli from collapsing. [13] PEEP is considered necessary with mechanical ventilation; however, if the pressure is too great it can expand the size of the contusion [17] and injure the lung. [39] When the compliance of the injured lung differs significantly from that of the uninjured one, the lungs can be ventilated independently with two ventilators in order to deliver air at different pressures; this helps avoid injury from overinflation while providing adequate ventilation. [58] Fluid therapy [ edit ] The administration of fluid therapy in individuals with pulmonary contusion is controversial. [41] Excessive fluid in the circulatory system ( hypervolemia ) can worsen hypoxia because it can cause fluid leakage from injured capillaries (pulmonary edema), which are more permeable than normal. [31] [43] However, low blood volume ( hypovolemia ) resulting from insufficient fluid has an even worse impact, potentially causing hypovolemic shock ; for people who have lost large amounts of blood, fluid resuscitation is necessary. [41] A lot of the evidence supporting the idea that fluids should be withheld from people with pulmonary contusion came from animal studies, not clinical trials with humans; human studies have had conflicting findings on whether fluid resuscitation worsens the condition. [20] Current recommendations suggest giving enough fluid to ensure sufficient blood flow but not giving any more fluid than necessary. [15] For people who do require large amounts of intravenous fluid, a catheter may be placed in the pulmonary artery to measure the pressure within it. [6] Measuring pulmonary artery pressure allows the clinician to give enough fluids to prevent shock without exacerbating edema. [59] Diuretics , drugs that increase urine output to reduce excessive fluid in the system, can be used when fluid overload does occur, as long as there is not a significant risk of shock. [15] Furosemide , a diuretic used in the treatment of pulmonary contusion, also relaxes the smooth muscle in the veins of the lungs, thereby decreasing pulmonary venous resistance and reducing the pressure in the pulmonary capillaries. [43] Supportive care [ edit ] Retaining secretions in the airways can worsen hypoxia [60] and lead to infections. [4] Thus, an important part of treatment is pulmonary toilet , the use of suction, deep breathing, coughing, and other methods to remove material such as mucus and blood from the airways. [7] Chest physical therapy makes use of techniques such as breathing exercises, stimulation of coughing, suctioning, percussion, movement, vibration, and drainage to rid the lungs of secretions, increase oxygenation, and expand collapsed parts of the lungs. [61] People with pulmonary contusion, especially those who do not respond well to other treatments, may be positioned with the uninjured lung lower than the injured one to improve oxygenation. [43] Inadequate pulmonary toilet can result in pneumonia. [40] People who do develop infections are given antibiotics. [17] No studies have yet shown a benefit of using antibiotics as a preventative measure before infection occurs, although some doctors do recommend prophylactic antibiotic use even without scientific evidence of its benefit. [13] However, this can cause the development of antibiotic resistant strains of bacteria, so giving antibiotics without a clear need is normally discouraged. [20] For people who are at especially high risk of developing infections, the sputum can be cultured to test for the presence of infection-causing bacteria; when they are present, antibiotics are used. [27] Pain control is another means to facilitate the elimination of secretions. A chest wall injury can make coughing painful, increasing the likelihood that secretions will accumulate in the airways. [62] Chest injuries also contribute to hypoventilation (inadequate breathing) because the chest wall movement involved in breathing adequately is painful. [62] [63] Insufficient expansion of the chest may lead to atelectasis , further reducing oxygenation of the blood. [35] Analgesics (pain medications) can be given to reduce pain. [12] Injection of anesthetics into nerves in the chest wall, called nerve blockade , is another approach to pain management; this does not depress respiration the way some pain medications can. [31] Prognosis [ edit ] This CT scan, taken 22 days after pulmonary contusion with major chest trauma, shows that the contusion has completely resolved. [64] Pulmonary contusion usually resolves itself [23] without causing permanent complications; [1] however it may also have long-term ill effects on respiratory function. [38] [65] Most contusions resolve in five to seven days after the injury. [1] Signs detectable by radiography are usually gone within 10 days after the injury—when they are not, other conditions, such as pneumonia, are the likely cause. [27] Chronic lung disease correlates with the size of the contusion and can interfere with an individual's ability to return to work. [24] Fibrosis of the lungs can occur, resulting in dyspnea (shortness of breath), low blood oxygenation, and reduced functional residual capacity for as long as six years after the injury. [38] As late as four years post-injury, decreased functional residual capacity has been found in most pulmonary contusion patients studied. [43] During the six months after pulmonary contusion, up to 90% of people suffer difficulty breathing. [27] [43] In some cases, dyspnea persists for an indefinite period. [7] Contusion can also permanently reduce the compliance of the lungs . [66] Complications [ edit ] A chest X-ray showing acute respiratory distress syndrome Pulmonary contusion can result in respiratory failure—about half of such cases occur within a few hours of the initial trauma. [43] Other severe complications, including infections and acute respiratory distress syndrome (ARDS) occur in up to half of cases. [31] Elderly people and those who have heart, lung, or kidney disease prior to the injury are more likely to stay longer in hospital and have complications from the injury.
-
Schizoaffective Disorder
Wikipedia
If mood disorder episodes are present for the majority and residual course of the illness and up until the diagnosis, the patient can be diagnosed with Schizoaffective Disorder. [5] Causes [ edit ] A combination of genetic and environmental factors are believed to play a role in the development of schizoaffective disorder. [14] [15] Genetic studies do not support the view that schizophrenia , psychotic mood disorders and schizoaffective disorder are distinct etiological entities, but rather the evidence suggests the existence of common inherited vulnerability that increases the risks for all these syndromes . Some susceptibility pathways may be specific for schizophrenia, others for bipolar disorder , and yet other mechanisms and genes may confer risk for mixed schizophrenic and affective [or mood disorder] psychoses, but there is no support from genetics for the view that these are distinct disorders with distinct etiologies and pathogenesis . ... Schizophrenia spectrum disorders, of which schizoaffective disorder is a part, have been increasingly linked to advanced paternal age at the time of conception, a known cause of genetic mutations. [16] The physiology of people diagnosed with schizoaffective disorder appears to be similar, but not identical, to that of those diagnosed with schizophrenia and bipolar disorder; however, human neurophysiological function in normal brain and mental disorder syndromes is not fully understood. [9] Substance abuse [ edit ] A clear causal connection between drug use and psychotic spectrum disorders, including schizoaffective disorder, has been difficult to prove. ... These may include serum BSL if olanzapine has previously been prescribed, thyroid function if lithium has previously been taken to rule out hypothyroidism , liver function tests if chlorpromazine has been prescribed, CPK levels to exclude neuroleptic malignant syndrome , and a urinalysis and serum toxicology screening if substance use is suspected. ... DSM-5 requires two episodes of psychosis (whereas DSM-IV needed only one) to qualify for the schizoaffective disorder diagnosis. [6] As such, it is no longer an "episode diagnosis." [6] The new schizoaffective framework looks at the time from "the [first episode of] psychosis up to the current episode [of psychosis], rather than only defining a single episode with [co-occurring] psychotic and mood syndromes ." [6] Specifically, one of the episodes of psychosis must last a minimum of two weeks without mood disorder symptoms, but the person may be mildly to moderately depressed while psychotic. [6] The other period of psychosis "requires the overlap of mood [disorder] symptoms with psychotic symptoms to be conspicuous" and last for a greater portion of the disorder. [41] These two changes are intended by the DSM-5 workgroup to accomplish two goals: [6] Increase the diagnosis' consistency (or reliability) when it is used; Significantly decrease the overall use of the schizoaffective disorder diagnosis. ... Psychiatric rehabilitation consists of eight main areas: Psychiatric (symptom reduction and management) Health and Medical (maintaining consistency of care) Housing (safe environments) Basic living skills ( hygiene , meals [including increasing healthy food intake and reducing processed food intake], safety, planning and chores) Social ( relationships , family boundaries, communication and integration of client into the community) Education and vocation (coping skills, motivation and suitable goals chosen by client) Finance ( personal budget ) Community and legal (resources) Medication [ edit ] Antipsychotic medication is usually required both for acute treatment and the prevention of relapse. [13] [59] There is no single antipsychotic of choice in treating schizoaffective disorder, but atypical antipsychotics should be considered because they have mood-stabilizing activity. [13] [56] Paliperidone is an antipsychotic with FDA approval for the treatment of schizoaffective disorder. [60] Antipsychotics should be used at the minimum dose necessary to control symptoms. [56] Potential side effects include extrapyramidal symptoms , including tremor , muscle stiffness, and restlessness or akathisia . [61] Atypical antipsychotics carry a risk of metabolic syndrome , including weight gain, increased blood sugar , and increased blood cholesterol , so regular monitoring of weight and blood work should be carried out. [61] Some atypical antipsychotics, such as ziprasidone and aripiprazole , are associated with less risk than others, such as olanzapine. [56] [61] Medication choice is based on how effectively it reduces symptoms, how few side effects it causes, and cost.GRIA1, BDNF, HTR2A, SULT4A1, SST, CALR, C9orf72, HFE, PCDH11Y, SIRT5, NEUROG1, NPY, NPAS2, PDGFRB, DRD5, PLA2G4A, ACSL6, SAT1, TF, TCF7L2, SNAP25, ERVW-4, AVP, HTR1B, CPLX1, CPLX2, NR3C1, GRIN1, PRKCA, MED15, NTRK1, SELENBP1, PVALB, RBM12, PLCB4, MAD1L1, SDCCAG8, RPN2, IPO8, LMO7, RGS6, ACTL7A, LINC02822, ITIH1, CCDC102B, SLC8A1-AS1, CABLES1, ATP6V1E2, TMPRSS5, LTN1, LINC02109, KSR2, UXS1, GIGYF1, STX2, CSMD1, ARHGEF10L, PCDH12, C11orf21, ZNF740, DISC1, COMT, DTNBP1, DRD3, DRD4, USH1G, TBX1, ADRA1A, DRD2, CYP2D6, DGCR, SSTR4, FOXP2, GAD1, ACKR3, MCCC2, GABRR1, EIF4EBP1, EGR3, EDNRA, DNTT, DLX4, DBH, DAO, SNRNP70, PPM1K, H19, CNTF, BRS3, SCZD12, LINC00273, BAG1, C20orf181, ECT, ARSA, AGA, GAD2, GPR42, GRID1, PCDH11X, SLC6A4, ADRA2B, THOP1, SAFB, RARB, LPAR2, ADIPOQ, PRODH, ABCB1, RTN3, SERPINE1, ARID3B, CXCR6, NOTCH4, NEUROD2, CD99, MAOA, CAPN10, HRH3, TBC1D9, KCNN3, IL18, IL1B, HRH1, NRG1, SLC1A1
-
Migraine
Wikipedia
Genetics [ edit ] Main article: Genetics of migraine headaches Studies of twins indicate a 34% to 51% genetic influence of likelihood to develop migraine. [3] This genetic relationship is stronger for migraine with aura than for migraines without aura. [27] A number of specific variants of genes increase the risk by a small to moderate amount. [53] Single gene disorders that result in migraines are rare. [53] One of these is known as familial hemiplegic migraine , a type of migraine with aura, which is inherited in an autosomal dominant fashion. [56] [57] Four genes have been shown to be involved in familial hemiplegic migraine. [58] Three of these genes are involved in ion transport . [58] The fourth is an axonal protein associated with the exocytosis complex. [58] Another genetic disorder associated with migraine is CADASIL syndrome or cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy. [2] One meta-analysis found a protective effect from an angiotensin converting enzyme polymorphisms on migraine. [59] The TRPM8 gene, which codes for a cation channel , has been linked to migraines. [60] Triggers [ edit ] Migraine may be induced by triggers, with some reporting it as an influence in a minority of cases [5] and others the majority. [61] Many things such as fatigue, certain foods, and weather have been labeled as triggers; however, the strength and significance of these relationships are uncertain. [61] [62] Most people with migraines report experiencing triggers. [63] Symptoms may start up to 24 hours after a trigger. [5] Physiological aspects [ edit ] Common triggers quoted are stress, hunger, and fatigue (these equally contribute to tension headaches ). [61] Psychological stress has been reported as a factor by 50 to 80% of people. [64] Migraine has also been associated with post-traumatic stress disorder and abuse. [65] Migraine episodes are more likely to occur around menstruation . [64] Other hormonal influences, such as menarche , oral contraceptive use, pregnancy , perimenopause, and menopause , also play a role. [66] These hormonal influences seem to play a greater role in migraine without aura. [54] Migraine episodes typically do not occur during the second and third trimesters of pregnancy, or following menopause. [2] Dietary aspects [ edit ] Between 12 and 60% of people report foods as triggers. [67] [68] Evidence for such triggers, however, mostly relies on self-reports and is not rigorous enough to prove or disprove any particular trigger. [69] A clear explanation for why food might trigger migraines is also lacking. [67] There does not appear to be evidence for an effect of tyramine – which is naturally present in chocolate, alcoholic beverages, most cheeses and processed meats – on migraine. [70] Likewise, while monosodium glutamate (MSG) is frequently reported, [71] evidence does not consistently support that it is a dietary trigger. [72] Environmental aspects [ edit ] A review on potential triggers in the indoor and outdoor environment concluded that there is insufficient evidence to confirm environmental factors as causing migraine. ... Now that this mechanism is not believed to be primary, the symptomatic term migraine with brainstem aura (MBA) is preferred. [46] Childhood periodic syndromes that are commonly precursors of migraine include cyclical vomiting (occasional intense periods of vomiting), abdominal migraine (abdominal pain, usually accompanied by nausea), and benign paroxysmal vertigo of childhood (occasional attacks of vertigo). ... Specifically, greater or equal to 15 days/month for longer than 3 months. [98] Abdominal migraine [ edit ] The diagnosis of abdominal migraine is controversial. [99] Some evidence indicates that recurrent episodes of abdominal pain in the absence of a headache may be a type of migraine [99] [100] or are at least a precursor to migraines. [27] These episodes of pain may or may not follow a migraine-like prodrome and typically last minutes to hours. [99] They often occur in those with either a personal or family history of typical migraine. [99] Other syndromes that are believed to be precursors include cyclical vomiting syndrome and benign paroxysmal vertigo of childhood . [27] Differential diagnosis [ edit ] Other conditions that can cause similar symptoms to a migraine headache include temporal arteritis , cluster headaches , acute glaucoma , meningitis and subarachnoid hemorrhage . [10] Temporal arteritis typically occurs in people over 50 years old and presents with tenderness over the temple , cluster headache presents with one-sided nose stuffiness, tears and severe pain around the orbits , acute glaucoma is associated with vision problems, meningitis with fevers , and subarachnoid hemorrhage with a very fast onset. [10] Tension headaches typically occur on both sides, are not pounding, and are less disabling. [10] Those with stable headaches that meet criteria for migraines should not receive neuroimaging to look for other intracranial disease. [101] [102] [103] This requires that other concerning findings such as papilledema (swelling of the optic disc) are not present.TRPM8, NOTCH3, PRDM16, LRP1, TGFBR2, SUGCT, FHL5, CALCA, ATP1A2, COL4A1, ESR1, HTR2A, GABRQ, HTR7, CACNA1A, MFN1, PRRT2, MEF2D, HTR1A, OPA1, COX2, SLC1A3, TGFB1, POLG, IL10, GABRG2, HPSE2, TREX1, PHACTR1, ASTN2, TWNK, CSNK1D, NRP1, PIK3CA, ADARB2, TRNS2, RPS27, RPS26, ZMYND8, RPS24, RPS20, RPS19, AMACR, RPS17, IL12A, RPS28, IL12B, INPP5A, KCNA1, RPS15A, RPS10, LEPROTL1, RRM2B, KCNQ2, KCNQ3, PRKG1, SRPX2, KRAS, HLA-B, MARCHF4, PMS1, MSH6, MYORG, FAN1, WAPL, MLH3, SLC24A3, SCN8A, PMS2, SCN2A, SCN1A, S100A12, PGK1, HTR1B, RPS29, PDGFB, NMUR2, WDR12, RELA, TRNS1, ND5, COX1, MYD88, COX3, CYTB, RPL5, MTHFR, MVK, ND1, ND4, ND6, NOS3, MRPL37, NEDD4L, TRNW, TRNF, TRNH, TRNV, TRNK, TRNL1, TRNQ, TRNC, NDP, MSH2, MMP17, RPS7, PLCE1, HJURP, RPL35A, NFIX, EPCAM, RPL27, SMAD4, RPL26, RPL18, NF2, MEFV, CNNM2, MLH1, GRIN2A, RPL15, ADA2, ERAP1, RPL11, SMAD3, SH2B1, RNF213, COL5A2, VHL, RNASEH1, ACSF3, VSTM4, CCR1, COL1A1, COL3A1, TWIST1, COL5A1, COMT, KCNK18, TNFRSF1A, CLVS1, IL23R, TNF, TLR4, YAP1, DBH, ACE, DRD2, UBAC2, SLC6A19, NOP56, PLCE1-AS1, ACVRL1, LINC02646, AIP, ADCYAP1, LINC02109, LINC01752, BAZ1B, IL12A-AS1, WASL, SLC25A4, MPPED2, PEX11B, APP, KCNK5, FAS, KLRC4, RABGAP1L, DNM1L, BMPR1A, C4A, CFDP1, MRVI1, GP1BB, GPR101, FBN2, SLC6A4, FGFR3, FGFR2, GDF2, GP1BA, RPL35, STIM1, CARF, SMARCB1, SUV39H2, POLG2, GATA2, SEMA4A, STAT4, GATA1, C11orf95, ADAMTSL1, ENG, NLRP3, GP9, MLX, NLRC4, HAUS1, DCLRE1C, SLC2A1, GCDH, TSR2, LTA, HCRT, BDNF, GOLPH3, TRPV1, APOE, TRPA1, MAOA, CRP, RAMP1, AVP, PSMG1, HTR1F, MZB1, LEP, DUSP2, VIP, IL6, ADCYAP1R1, KLF6, PGR, SLC6A3, PTGS2, GRIA1, ADIPOQ, GRIA3, CNR1, F5, NOS1, EDNRA, HTR2C, TAC1, MTDH, SPRTN, INSR, NOS2, PANX1, PTX3, STX1A, POMC, VEGFA, CALCRL, VWF, TSPAN2, STIN2-VNTR, DRD3, HTR1D, P2RX7, AGT, KCNN3, HLA-DRB1, OPRM1, FAAH, HCRTR1, DAO, GC, SOD2, FMR1, HCRTR2, GRIN2B, MGLL, HNMT, UTS2, PART1, EDN1, TPH2, ICAM1, CYP19A1, RLS1, TYRP1, TSPAN33, CACNA1C, MIR34A, HEPH, ALB, MTCO2P12, ASIC3, IAPP, GABRA3, TBPL1, REN, MTHFD1, PRL, KCNK2, PON1, AKR1A1, CD274, GABRR3, MMD, ADHD1, TRIM13, ANKK1, MGR6, PYCARD, PTCRA, CCL4L1, MIR30A, TRPV3, REM1, MIR375, NR1I3, PSAT1, CCL4L2, LOC110806262, LOC107987479, NBEA, ACAD8, IL37, OPN1MW3, LINC01672, CCR2, KLK4, BMS1, OPN1MW2, CXADRP1, MIR382, BACE1, OR2AG1, AJAP1, SYNE1, RETN, P2RY12, ACE2, PGPEP1, RPTOR, MOCOS, NPEPPS, OLAH, ZMIZ1, CHMP1B, IMPACT, VWA8, TBC1D9, PAG1, ZKSCAN7, ZC4H2, KCNK10, HRH3, MAPKAP1, ATP5MD, RBM45, GAL, TRAF3IP2, EFHC1, OPN4, DCLK3, NES, CA14, CSAD, CYSLTR1, HPSE, ISYNA1, ADRM1, ACSL5, AK3, AOC1, ROCK2, F9, GABRR2, GABRR1, FSHR, FOS, FLNA, FOXM1, F11, F2RL1, TNFSF10, F2, ETFA, ESR2, ESD, ERCC5, EPHB2, EDNRB, GAD1, GCH1, OPN1MW, GEM, IFNG, HTR5A, HLA-DQB2, HLA-DQB1, HFE, GUCY2D, GSTM2, GSTM1, GRIA4, GRIA2, UTS2R, GPI, GNB3, GNAS, GNAO1, DRD5, DRD4, CYP2D6, OPN1SW, STS, ARR3, AQP4, KLK3, APEX1, ANK3, AMELX, AKT1, AGTR1, ADRA2B, ADH1B, ADARB1, ADAR, ACHE, ASIC1, ATP1A3, BGN, CYP1A1, C3, CXADR, CX3CR1, CTLA4, COL4A2, CLTA, CES1, CD40LG, CD40, ENTPD1, CD14, CAV2, CAT, CASR, CALCR, CACNA1E, IGF1, IL1A, IL1B, SPTAN1, SPAST, SOD1, SLCO1A2, SLC20A2, SLC1A2, SELL, CCL4, CCL2, S100B, RYR2, RNASE2, RHAG, PTPRC, PTEN, PROS1, SPG7, SST, MAPK1, STATH, SLC4A4, NR4A3, XRCC3, XRCC1, WAS, VIPR2, VIPR1, VDR, TYMS, TPH1, TNFRSF1B, TIMP2, THAS, TH, TGFBI, PRNP, ACACA, IL1RN, MUC1, MTR, MMP16, MMP9, MMP3, MMP2, MEIS1, SMCP, MAOB, LPA, LGALS3, LDLR, LBR, TNPO1, IL9, CXCL8, MTRR, NGF, PNOC, NNMT, PLAG1, PIK3CG, PIK3CD, PIK3CB, ABCB1, PRDX1, P2RX4, P2RX3, OPRK1, OGG1, NTSR1, NT5E, NRAS, NOTCH4, NPY, PRKAR1A
-
Dehydrated Hereditary Stomatocytosis 1 With Or Without Pseudohyperkalemia And/or Perinatal Edema
Omim
See 137280 for a discussion of the association of familial stomatocytosis and hypertrophic gastritis in the dog, an autosomal recessive syndrome. Reviews Delaunay (2004) reviewed genetic disorders of red cell membrane permeability to monovalent cations, noting 'inevitable' overlap between entities based on clinical phenotype. ... Grootenboer et al. (1998) described a pleiotropic, autosomal dominant syndrome consisting of DHS, hereditary pseudohyperkalemia, and severe perinatal edema, including ascites. ... Rees et al. (2004) noted that 3 families had been reported in which dehydrated hereditary stomatocytosis was associated with a syndrome of self-limiting perinatal ascites (Entezami et al., 1996; Grootenboer et al., 2000; Basu et al., 2003). ... Grootenboer et al. (2000) presented evidence that DHS with pseudohyperkalemia and perinatal edema is a pleiotropic syndrome in which some features may be missing.
-
Dementia With Lewy Bodies
Wikipedia
It is one of the two Lewy body dementias , along with Parkinson's disease dementia. [13] The atypical parkinsonian syndromes include DLB, along with other conditions. [14] Lastly, DLB is a synucleinopathy , meaning that it is characterized by abnormal deposits of alpha-synuclein protein in the brain. ... The differential diagnosis includes Alzheimer's disease; such synucleinopathies as Parkinson's disease dementia, Parkinson's disease, and multiple system atrophy; vascular dementia; and progressive supranuclear palsy , corticobasal degeneration , and corticobasal syndrome . [5] The symptoms of DLB are easily confused with delirium, [122] or more rarely as psychosis; [103] prodromal subtypes of delirium-onset DLB and psychiatric-onset DLB have been proposed. [20] Mismanagement of delirium is a particular concern because of the risks to people with DLB associated with antipsychotics. [122] A careful examination for features of DLB is warranted in individuals with unexplained delirium. [123] PET or SPECT imaging showing reduced dopamine transporter uptake can help distinguish DLB from delirium. [122] Lewy pathology affects the peripheral autonomic nervous system; autonomic dysfunction is observed less often in AD, frontotemporal, or vascular dementias, so its presence can help differentiate them. [124] MRI scans almost always show abnormalities in the brains of people with vascular dementia, which can begin suddenly. [125] Alzheimer's disease [ edit ] DLB is distinguishable from AD even in the prodromal phase. [21] Short-term memory impairment is seen early in AD and is a prominent feature, while fluctuating attention is uncommon; impairment in DLB is more often seen first as fluctuating cognition. [126] In contrast to AD—in which the hippocampus is among the first brain structures affected, and episodic memory loss related to encoding of memories is typically the earliest symptom—memory impairment occurs later in DLB. [31] [127] People with amnestic mild cognitive impairment (in which memory loss is the main symptom) may progress to AD, whereas those with non-amnestic mild cognitive impairment (which has more prominent impairments in language, visuospatial, and executive domains) are more likely to progress towards DLB. [128] Memory loss in DLB has a different progression from AD because frontal structures are involved earlier, with later involvement of temporoparietal brain structures. [127] Verbal memory is not as severely affected as in AD. [129] While 74% of people with autopsy-confirmed DLB had deficits in planning and organization, they show up in only 45% of people with AD. [130] Visuospatial processing deficits are present in most individuals with DLB, [57] and they show up earlier and are more pronounced than in AD. [131] Hallucinations typically occur early in the course of DLB, [5] are less common in early AD, but usually occur later in AD. [76] AD pathology frequently co-occurs in DLB, so the cerebrospinal fluid (CSF) testing for Aβ and tau protein that is often used to detect AD is not useful in differentiating AD and DLB. [49] PET or SPECT imaging can be used to detect reduced dopamine transporter uptake and distinguish AD from DLB. [49] [132] Severe atrophy of the hippocampus is more typical of AD than DLB. [133] Before dementia develops (during the mild cognitive impairment phase), MRI scans show normal hippocampal volume. After dementia develops, MRI shows more atrophy among individuals with AD, and a slower reduction in volume over time among people with DLB than those with AD. [26] Compared to people with AD, FDG-PET brain scans in people with DLB often show a cingulate island sign . [26] In East Asia, particularly Japan, 123 I-MIBG is used in the differential diagnosis of DLB and AD, because reduced labeling of cardiac nerves is seen only in Lewy body disorders. [103] [120] Other indicative and supportive biomarkers are useful in distinguishing DLB and AD (preservation of medial temporal lobe structures, reduced occipital activity, and slow-wave EEG activity). [24] Synucleinopathies [ edit ] Dementia with Lewy bodies and Parkinson's disease dementia are clinically similar after dementia occurs in Parkinson's disease. [134] Delusions in Parkinson's disease dementia are less common than in DLB, [135] and persons with Parkinson's disease are typically less caught up in their visual hallucinations than those with DLB. [76] There is a lower incidence of tremor at rest in DLB than in Parkinson's disease, and signs of parkinsonism in DLB are more symmetrical. [34] In multiple system atrophy, autonomic dysfunction appears earlier and is more severe, [31] and is accompanied by uncoordinated movements, while visual hallucinations and fluctuating cognition are less common than in DLB. [136] Urinary difficulty is one of the earliest symptoms with multiple system atrophy, and is often severe. [61] Frontotemporal dementias [ edit ] Corticobasal syndrome, corticobasal degeneration and progressive supranuclear palsy are frontotemporal dementias [137] with features of parkinsonism and impaired cognition. Similar to DLB, imaging may show reduced dopamine transporter uptake. Corticobasal syndrome and degeneration, and progressive supranuclear palsy, are usually distinguished from DLB by history and examination. Motor movements in corticobasal syndrome are asymmetrical. There are differences in posture, gaze and facial expressions in the most common variants of progressive supranuclear palsy, and falling backwards is more common relative to DLB.SNCA, SNCB, GBA, TH, NGF, GFAP, SOD2, ENO2, EDN1, IGF2, IGF2R, INS, PPARGC1A, NTRK1, NTRK2, INSR, MAG, BECN1, MAP2, IGF1R, AIF1, AGER, APOE, ASH1L, TARDBP, CSF2, LRRK2, LAMC2, PSEN1, MAPT, BCL7C, MIR762HG, MIR4519, BCHE, SLC6A3, CIT, CYP2D6, APP, SMUG1, SNCG, GBAP1, MCIDAS, IMPA1, BRAP, GRN, SNCAIP, PRNP, LRP10, NOS3, TPO, ACHE, BDNF, CHI3L1, CTSD, IGF1, SYNM, HSPA4, C9orf72, ACE, CYP2D7, PSMB8, SERPINA3, KLK6, PLA2G6, TSHZ1, TOMM40, SCARB2, NEFL, PRKN, NOS2, ZFPM1, GRM5, CALB1, PINK1, EIF4G1, REM1, NUB1, FYN, SPP1, MIR34B, ATG7, RIDA, DLEU1, VCAM1, UCHL3, UCHL1, KHDRBS1, OGA, UBC, PLK2, UBB, ALDH1L1, PHYHD1, NANOS3, ECE2, MIR451A, AKR7A2, MIR21, MIR133B, SUCLA2, VSNL1, SQSTM1, AD12, TFEB, PPIG, MTCO2P12, VGF, GDF15, LOC107987479, VEGFA, MYO5C, CHP1, PTCRA, BHLHE22, NBEAL1, TPSG1, CHMP2B, MOAP1, NEUROD6, TIGAR, CHORDC1, GPR78, HTRA2, TSC2, TMEM230, RMC1, GAL, CHCHD2, DCTN4, ABI3, SLC17A7, VPS13C, NUP62, MUL1, SUGT1, BPIFA2, TPPP, CBLL2, LAPTM4B, PARK7, ECD, SESN3, SYNPO, LMTK2, RPH3A, MINDY4, SMG1, KCNH8, DNAJC13, ZFPM2, RAB39B, TRIM9, KCNH4, CHRFAM7A, BACE1, A2M, TNF, DBH, DLD, DLG4, DNMT1, DRD1, DRD2, DYRK1A, ECE1, EEF1A1, EEF1B2P2, ELF3, ELK1, EPHX2, F11, FABP3, FANCD2, FGFR3, FKBP1A, FKBP1AP1, FKBP1AP2, FKBP1AP3, FKBP1AP4, DCX, CYP2B6, FUS, CTSB, GRK2, AFM, ANK1, BIRC5, AQP1, AQP4, ASAH1, BCL2, BCL2L1, BMI1, BRCA2, TSPO, CAMK2A, CAPN1, CASP2, CASP3, CHRNA4, CLU, COMT, CSF1R, CST3, FOXO1, SFN, TGFB2, SERPINB6, PLCG2, PRD, PSPN, PRSS2, MASP1, PSEN2, PSPH, PTGS2, REG1A, REST, RPS27A, S100B, SGTA, SLC1A2, SLC6A2, SLC6A4, SOD1, SP1, STXBP3, TF, TFAM, PIK3R2, SERPINA1, GRK5, PECAM1, GSN, GTF2H1, HSP90AA1, IGFALS, IL6, INSRR, ITGB2, LAD1, LAMP2, LGALS3, MAOB, MAP1A, AFF1, MMP9, MSMB, COX2, NOS1, NOTCH3, NRGN, NR4A2, PDHA1, EEF1AKMT4-ECE2
-
Psychosis
Wikipedia
In addition, diagnostic criteria include at least four of the following symptoms: [38] Confusion Mood-incongruent delusions Hallucinations Pan-anxiety, a severe anxiety not bound to particular situations or circumstances Happiness or ecstasy of high degree Motility disturbances of akinetic or hyperkinetic type Concern with death Mood swings to some degree, but less than what is needed for diagnosis of an affective disorder Cycloid psychosis occurs in people of generally 15–50 years of age. [38] Medical conditions [ edit ] A very large number of medical conditions can cause psychosis, sometimes called secondary psychosis . [24] Examples include: disorders causing delirium ( toxic psychosis ), in which consciousness is disturbed neurodevelopmental disorders and chromosomal abnormalities, including velocardiofacial syndrome neurodegenerative disorders, such as Alzheimer's disease , [39] dementia with Lewy bodies , [40] and Parkinson's disease [41] [42] focal neurological disease, such as stroke , brain tumors , [43] multiple sclerosis , [42] and some forms of epilepsy malignancy (typically via masses in the brain, paraneoplastic syndromes ) [42] infectious and postinfectious syndromes, including infections causing delirium , viral encephalitis , HIV/AIDS , [44] malaria , [45] syphilis [46] endocrine disease, such as hypothyroidism , hyperthyroidism , Cushing's syndrome , hypoparathyroidism and hyperparathyroidism ; [47] sex hormones also affect psychotic symptoms and sometimes giving birth can provoke psychosis, termed postpartum psychosis [48] inborn errors of metabolism, such as Succinic semialdehyde dehydrogenase deficiency , porphyria and metachromatic leukodystrophy [49] [50] [51] [52] nutritional deficiency, such as vitamin B 12 deficiency [9] other acquired metabolic disorders, including electrolyte disturbances such as hypocalcemia , hypernatremia , [53] hyponatremia , [54] hypokalemia , [55] hypomagnesemia , [56] hypermagnesemia , [57] hypercalcemia , [58] and hypophosphatemia , [59] but also hypoglycemia , [60] hypoxia , and failure of the liver or kidneys autoimmune and related disorders, such as systemic lupus erythematosus (lupus, SLE), sarcoidosis , Hashimoto's encephalopathy , anti-NMDA-receptor encephalitis , and non-celiac gluten sensitivity [49] [61] poisoning, by therapeutic drugs (see below), recreational drugs (see below), and a range of plants, fungi, metals, organic compounds, and a few animal toxins [24] sleep disorders, such as in narcolepsy (in which REM sleep intrudes into wakefulness) [24] parasitic diseases, such as neurocysticercosis Psychoactive drugs [ edit ] Main article: Substance-induced psychosis Various psychoactive substances (both legal and illegal) have been implicated in causing, exacerbating, or precipitating psychotic states or disorders in users, with varying levels of evidence. ... People on typical antipsychotics tend to have a higher rate of extrapyramidal side effects while some atypicals are associated with considerable weight gain, diabetes and risk of metabolic syndrome ; this is most pronounced with olanzapine, while risperidone and quetiapine are also associated with weight gain. [123] Risperidone has a similar rate of extrapyramidal symptoms to haloperidol. [123] Counseling [ edit ] Psychological treatments such as acceptance and commitment therapy (ACT) are possibly useful in the treatment of psychosis, helping people to focus more on what they can do in terms of valued life directions despite challenging symptomology. [129] There are psychological interventions that seek to treat the symptoms of psychosis.CACNA1C, C9orf72, MED12, PAH, BDNF, HTR2A, PDE10A, LEP, FKBP5, MAOA, ANK3, GCH1, NR3C1, CALR, TCF4, S100B, SERPINA3, PVALB, KCNH2, CYP2B6, CRHR1, NPAS3, ADH1B, TET1, NDEL1, GRIN1, DLX1, TPH2, PICK1, FEZ1, RAPGEF5, OPRM1, NPY1R, RBM12, SHOX2, OLIG2, SEPTIN2, FTL, NQO2, GADD45B, WWC1, SAT1, TENM4, DRD5, APOBEC3C, SLC39A3, SHANK1, CARTPT, SP3, TBX1, ST3GAL1, PLAT, ABCB1, TDO2, HTR5A, TGFB1, SLC6A9, TRPM2, DDC, TYR, GPR52, GSTP1, FZD3, PLA2G6, ME2, SELENBP1, PTPRC, GRIK4, GRIK3, GLUL, TOMM40, DBI, NDE1, CSNK1E, KCNH6, APOBEC3A, SP8, GNB1L, SOBP, PCDH11Y, PBRM1, CNP, CYP27B1, ZSCAN31, CD34, PAK5, CCKAR, ST6GALNAC1, SP1, GSTA1, GNA12, PPP3CC, HDAC2, CSF2RB, CDH17, PCNT, FGFR2, PRKCA, CPLX2, HTR1B, BCL9, NPAS1, SNAP25, DGCR8, HPGDS, HLA-E, NTRK1, LMOD2, SLC12A2, ADARB1, MEF2A, HLA-B, ARHGAP26, PAWR, AVP, CPLX1, MECP2, MAPT, RPS6KA3, PRNP, PCDH19, TWNK, GRN, SLC12A6, PGPEP1, PAK3, PDGFRB, NPC1, SLC20A2, LINC01602, CLDN11, TMEM182, LINC00243, DIPK1A, SAA2-SAA4, PDGFB, ACADS, TRIM9, NALCN-AS1, ATXN7, PRKAR1A, SAA2, NHLRC1, NCKAP5, ALDH18A1, PIK3C2A, NALCN, SLC6A19, DAOA, PSEN1, PPOX, TTLL6, PRL, ZNF804A, WFS1, SHANK3, SPP1, TTC19, SLC25A13, KCNMB2, SPTLC1, NPC2, TMEM106B, TREX1, SPART, PHF8, VPS13A, ZFYVE26, SH2B1, CHMP2B, CPVL, CNTNAP2, PTPN22, DISC1, RIN2, PDE11A, TREM2, ASCC1, LMBRD1, ITM2B, PRDM8, LRP8, STAT4, PGBD1, COL21A1, DCAF17, PANK2, VCP, VRK2, ZDHHC9, UPF3B, ALDH5A1, PLAA, EPM2A, MAD1L1, ADAM19, ARHGEF7, SQSTM1, CDH23, SLC24A3, USP8, TAOK2, NFKB1, SLC6A4, H3P40, DNASE1, C1R, CACNA1A, CBS, CD40, CHN2, IRAK1, CLN3, COMT, CRP, CTLA4, DRD2, BPI, DRD3, DRD4, CLN8, FCGR2A, FCGR2B, NDP, GCLC, GNAS, HLA-A, HFE, C1QA, NRG1, APOE, ALAD, ABCD1, AMPD3, AKT1, IL6, DTNBP1, GAD1, TNF, DRD1, TSPO, SMC2, KIDINS220, DNASE1L3, CNR1, GABRB2, SLC6A3, MIR137, GRM2, GRM5, IL1B, DAO, NHS, CYP2D6, RELN, NRGN, KMO, HTR2C, POTEM, FGF9, ACTG2, ACOT7, POTEKP, GRIN2B, MTHFR, GRM3, FHL5, ACTBL2, PAFAH1B1, ACTG1, OPN1SW, RGS4, VEGFA, LGI1, GLP1R, ECT, GRIA2, VDR, EBPL, GRIN2A, YWHAE, WAS, GSTT1, CHRFAM7A, LINC00273, RGS9, NAT8L, ADAMTS2, CLINT1, ALDH2, CHRNA7, SCLY, CHRNA4, DNMT1, CHPT1, TNRC6A, CHI3L1, RTN4R, CDT1, SF3B1, ELK3, ERBB4, CCK, DHDDS, TET2, ASXL1, BAG1, GAD2, RTN3, GABRA1, SAFB, CCL2, KRT7, POMC, SOD1, SOD2, HTR1A, INSRR, SERPINE1, MSH3, TAL1, CCL11, IFNG, RARB, THOP1, PYCR1, ISG20, ACE2, RASA1, IL2RA, CDK9, KCNN3, CD9, IL2, CSMD1, PPARG, FTO, MCPH1, ALG9, EHMT1, PLG, KRAS, PLA2G4A, PLA2G1B, NPL, DNMT3B, LCN1, LEPR, LGALS3, BMP1, CHGB, PPT1, IL6R, PRCP, CCHCR1, CYBB, CUX1, PDP1, CXCL8, AHI1, DYM, PTPN4, IL10, DAPK3, PRSS2, SLC52A1, CNTF, MTPAP, DBH, IL17A, IL18, TLR9, MAPK4, TMPRSS13, RETN, CHRM3, SNX7, PRKCD, ZGPAT, PDE4D, PPP1R1B, MSMB, ALAS2, TNFRSF11B, NTS, ZFP57, LRRTM1, GSTK1, NTRK2, AHR, ADCY5, MST1, MIR223, ND4, ND5, MIR338, NOTCH4, NOTCH2, ZGLP1, C20orf181, SCZD13, NPY, MTNR1B, ACP3, ACHE, LINC02210-CRHR1, STIN2-VNTR, H3P19, CXCL17, OXTR, MMP9, CSMD2, LRPAP1, DPYSL2, SERPINA5, BCHE, LMLN, MAOB, KCNH7, IL33, CREB3L1, REG3A, ATP2A2, TIRAP, CGAS, KLK3, LRRK2, ARSB, NAXE, ARNTL, SLCO6A1, TAAR1, NRSN1, PPM1K, PRICKLE2, MGAT1, ARID2, AQP4, ASAP1, CLDN5, PYCARD, REM1, PER2, STAT1, HSPA9, SKAP2, CHRNA6, SPAST, KALRN, CCRL2, GRIA3, PCSK7, GLP2R, GRIA1, ADIPOQ, NRXN1, HTR6, SMS, SMARCA1, TBX4, SLC22A3, GDF15, GHR, RGS6, GH1, HTR7, GFAP, GAB2, GCG, NR1I2, ASAP2, NRP1, HNF1A, TPH1, TXN, TYMS, TH, UBE3A, UROD, VCAM1, HBA2, TGIF1, TGFB2, HAL, UBE2K, HLA-C, HRH1, ZNF41, GSTM5, GSTM1, GSK3B, GRM8, SLC7A5, TRRAP, GRM7, TBCA, CPZ, HLA-DRB1, KCNK5, SLC18A2, SLC12A3, GABRG3, CNRIP1, NMNAT2, IGFALS, F2RL1, SHBG, SEMG1, SELE, TARDBP, EGR1, SMUG1, PDSS1, IBGC1, BRD1, EEF1A2, SV2C, EDA, E2F1, DRP2, B3GAT1, EIF2AK1, DISC2, ATXN8OS, IGFBP2, IL1A, PCDH11X, TP53, SGSM3, SPEN, SEPHS1, ALYREF, SLC1A1, GABRG2, HCG9, IGF1, IGF1R, PITRM1, G6PD, ARFGEF1, FMR1, SLC1A2, SH2B2, ARID3B, FGFR3, FES, TUSC2, MRPS30, PAPOLA, TMED2, SLC27A5, TPPP, WDHD1, HRH3, CHP1, SLAMF1, SCN11A, IGF2, PDAP1, NF2
-
Iron Overload
Wikipedia
The most important causes are hereditary haemochromatosis (HHC), a genetic disorder, and transfusional iron overload , which can result from repeated blood transfusions . [1] Contents 1 Signs and symptoms 2 Causes 2.1 Primary haemochromatosis 2.2 Secondary haemochromatosis 3 Diagnosis 4 Treatment 4.1 Chelating polymers 5 Prognosis 6 Epidemiology 7 History 7.1 Stone Age 7.2 Viking hypothesis 7.3 Modern times 8 Terminology 9 See also 10 References 11 External links Signs and symptoms [ edit ] Organs most commonly affected by haemochromatosis are the liver , heart , and endocrine glands . [2] Haemochromatosis may present with the following clinical syndromes: [3] Chronic liver disease and cirrhosis of the liver Heart involvement: heart failure , irregular heart rhythm Hormonal issues: diabetes (see below) and hypogonadism (insufficiency of the sex hormone producing glands) which leads to low sex drive and/or loss of fertility in men and loss of menstrual cycle in women Diabetes in people with iron overload occurs as a result of selective iron deposition in islet beta cells in the pancreas leading to functional failure and cell death. [4] Arthritis , from calcium pyrophosphate deposition in joints leading to joint pains. ... Primary haemochromatosis [ edit ] Although it was known most of the 20th century that most cases of haemochromatosis were inherited, they were incorrectly assumed to depend on a single gene. [9] The overwhelming majority depend on mutations of the HFE gene discovered in 1996, but since then others have been discovered and sometimes are grouped together as "non-classical hereditary haemochromatosis", [10] "non-HFE related hereditary haemochromatosis", [11] or "non-HFE haemochromatosis". [12] Description OMIM Mutation Haemochromatosis type 1 : "classical" haemochromatosis 235200 HFE Haemochromatosis type 2A : juvenile haemochromatosis 602390 Haemojuvelin (HJV, also known as RGMc and HFE2) Haemochromatosis type 2B : juvenile haemochromatosis 606464 hepcidin antimicrobial peptide ( HAMP ) or HFE2B Haemochromatosis type 3 604250 transferrin receptor-2 (TFR2 or HFE3) Haemochromatosis type 4 / African iron overload 604653 ferroportin (SLC11A3/SLC40A1) Neonatal haemochromatosis 231100 (unknown) Acaeruloplasminaemia (very rare) 604290 caeruloplasmin Congenital atransferrinaemia (very rare) 209300 transferrin GRACILE syndrome (very rare) 603358 BCS1L Most types of hereditary haemochromatosis have autosomal recessive inheritance, while type 4 has autosomal dominant inheritance. [13] Secondary haemochromatosis [ edit ] Severe chronic haemolysis of any cause, including intravascular haemolysis and ineffective erythropoiesis (haemolysis within the bone marrow ) Multiple frequent blood transfusions (either whole blood or just red blood cells ), which are usually needed either by individuals with hereditary anaemias (such as beta-thalassaemia major , sickle cell anaemia , and Diamond–Blackfan anaemia ) or by older patients with severe acquired anaemias such as in myelodysplastic syndromes . [14] Excess parenteral iron supplements, such as what can acutely happen in iron poisoning Excess dietary iron Some disorders do not normally cause haemochromatosis on their own, but may do so in the presence of other predisposing factors. ... Iron overload at Curlie GeneReview/NCBI/NIH/UW entry on HFE-Associated Hereditary Hemochromatosis GeneReview/NCBI/NIH/UW entry on TFR2-Related Hereditary Hemochromatosis GeneReview/NCBI/NIH/UW entry on Juvenile Hereditary Hemochromatosis GeneReview/NCBI/NIH/UW entry on Aceruloplasminemia v t e Metal deficiency and toxicity disorders Iron excess: Iron overload Hemochromatosis Hemochromatosis/HFE1 Juvenile/HFE2 HFE3 African iron overload/HFE4 Aceruloplasminemia Atransferrinemia Hemosiderosis deficiency: Iron deficiency Copper excess: Copper toxicity Wilson's disease deficiency: Copper deficiency Menkes disease / Occipital horn syndrome Zinc excess: Zinc toxicity deficiency: Acrodermatitis enteropathica Other Inborn errors of metabolismHAMP, CP, SLC40A1, BMP6, FTH1, CYBRD1, TF, IREB2, MAPT, ATP2A2, ENTPD1, GGT1, SLC11A2, CHMP2B, HFE, TFR2, TFRC, DMRT1, FXN, HJV, ALAS2, TMPRSS6, EPO, ERFE, UROD, ROS1, IL6, ADIPOQ, EPAS1, NFE2L2, HEPH, FTL, CRP, PGF, ABCB7, HP, HLA-A, GABPA, HMOX1, STAT3, CDAN1, FBXL5, SNCA, CAT, APP, B2M, GDF15, CCL2, ICAM1, HBB, MIR122, HIF1A, SF3B1, DNM1L, PIEZO1, NCOA4, GNPAT, PIK3CG, TP53, TNF, TGFB1, LCN2, GPX4, SLC11A1, PIK3CA, PIK3CB, PIK3CD, SHBG, GSTM1, MAPK8, RN7SL263P, CASP3, COL11A2, APOE, PARP1, RUNX2, RNF19A, SIRT1, SEC23B, AHSA1, CCL27, POLDIP2, WDR45, CUL9, ATRNL1, PRDX5, ISCU, SLC39A14, AQP4, DAPK2, APOC3, DIANPH, GDF11, CASP8AP2, ABCB6, APC, AIMP2, BDNF, BCS1L, DENR, TP63, SOCS1, EIF3A, APLN, PER2, PCSK7, BCL2L1, GRAP2, BCL2, TBPL1, CCL4L2, BAX, ATHS, PTPN22, SLC17A5, VEGFA, AHR, ASRGL1, SESN2, REPS1, GGTLC1, FTMT, SFXN1, SPNS2, SLCO6A1, AGTR1, HAVCR1, AGT, GSTK1, CCL4L1, ACVR1, MIR374A, BOLA2, MIR455, LOC102724197, AKT1, HHIPL2, PINK1, NOD2, PDLIM3, HPGDS, ALPP, NOX4, TRNT1, GLRX5, SLC25A37, PRRX2, UGT1A1, SLC25A38, NAT10, STAB2, SUCNR1, BDH2, MFF, MCOLN1, ALOX5, TRPV1, GPT, UTRN, LEP, SMAD4, SMAD6, SMAD7, SMAD9, DGUOK, MPO, COX2, MUC3A, MUC7, MYD88, NEO1, DES, NGF, NMBR, NPPB, DECR1, PAEP, LRPAP1, NQO1, SERPINA1, IRS1, HBA1, HBA2, GPER1, HTT, GPI, G6PD, MTOR, HLA-B, FOXO3, FDXR, HPX, FANCC, F3, CXCL8, IL10, CXCL10, EPHB2, DDIT3, PIGA, GSTM2, SPP1, CD59, STAT5A, STAT5B, SYP, ADAM17, TBXAS1, TDGF1P3, PRDX2, TERF2, CD38, CBS, CASR, TIMP3, TLR2, TLR4, BMPR1A, BGLAP, SPRR2A, SOD2, CYBB, CD68, CTNNB1, MAPK14, CRMP1, PRB1, PREP, MAPK1, ACO1, REN, CRK, RPE, SERPINB3, SCT, CREB1, CCL4, CCL18, CHIT1, SLPI, USF2
-
Cerebral Cavernous Malformations
Omim
They recommended that any patient with a vascular malformation, especially a cavernous one of the brain, spinal cord, or retina, be evaluated for the possibility of this syndrome, which they referred to as 'familial cavernous malformations of CNS and retina' (FCMCR). ... Corboy and Galetta (1989) described a family in which the proband had suffered for 9 years from recurrent 'acute chiasmal syndrome,' diagnosed at first as retrobulbar neuritis. ... The patient's mother and 6-year-old son had macular hemangiomas of the face and trunk and her brother had classic Klippel-Trenaunay-Weber syndrome (149000) of the right lower extremity. ... The chromosomal location distinguishes this disorder from the autosomal dominant vascular malformation syndrome (VMCM; 600195) in which lesions are primarily cutaneous; VMCM is due to mutation in a gene that maps to 9p21.
-
Wilson Disease
Gene_reviews
Low-molecular weight proteinuria, microscopic hematuria, and Fanconi syndrome Arthritis. Involvement of large joints from synovial copper accumulation Reduced bone mineral density with a prevalence of osteoporosis in approximately 10% of affected individuals Pancreatitis, cardiomyopathy, cardiac arrhythmias, rhabdomyolysis of skeletal muscle, and various endocrine disorders Sunflower cataracts observed occasionally on slit lamp examination Hepatocellular carcinoma rarely develops in Wilson disease: the estimated incidence is below 1% [Devarbhavi et al 2012]. ... Decreased serum concentrations of ceruloplasmin are observed in protein-losing enteropathy, nephrotic syndrome, and malnutrition, but also in some heterozygotes for Wilson disease. ... Serious side effects can occur in up to 30% of individuals, and include: severe thrombocytopenia, leukopenia, aplastic anemia, proteinuria, nephrotic syndrome, polyserositis, Goodpasture syndrome, and severe skin reactions.ATP7B, ANXA5, CP, APOE, PRNP, IL6, AHCY, LOX, TNF, IL10, LOXL2, A2M, SNCA, NDUFB7, PPP3CB, PPP3CA, CXCL8, CAMK2A, TIMP1, HAMP, ASMT, SDHAF2, BHMT, ANKS1B, ATP7A, CYP27B1, HMGCR, SMPD1, ESD, ATOX1, COMMD1, HFE, MBD6, ALB, TP53, SLC31A1, GPT, MBD1, PANK2, SERPINA1, PNOC, STON1, IL1B, GCH1, GTF2A1L, STON1-GTF2A1L, AFM, G6PD, BDNF, FXN, ARSA, GLUL, BMS1, SLC39A14, TBC1D9, FNDC3A, PDCD10, ATHS, CCL27, AHSA1, CCS, XPR1, GRAP2, CHMP2B, PRC1, AIMP2, XRCC5, BEST1, VEGFA, VCAM1, TYR, TXN, RNF19A, SLC17A5, ATRNL1, IGAN1, ACACA, CCDC115, PNPLA3, DNAJC5, ASRGL1, ALPP, PAGR1, SLC39A8, CPAT1, RBPJP4, POLDIP2, AMELX, XIAP, NAT10, INO80, IL23A, GOLM1, ZDHHC2, PDLIM3, CALCR, BRAF, CAT, GALNS, THRB, MT1X, ACE, LTA, DDIT3, DMD, KDR, IDO1, IL13, DMRT1, DNAH8, DRD2, IL2, IL1RN, IL1R1, IL1A, HNF4A, FOXA2, FOXA1, GAST, GRM5, GPX1, MTF1, MTHFR, SERPINA3, MAPK8, SPG7, SOD2, CFTR, CLU, SLPI, RXRA, RB1, PROP1, SLC31A2, MAPK1, DBH, CRK, CRP, PPARG, MAPK14, CTNNB1, ABCB1, PAH, OPA1, SLC11A2, NDUFAB1
-
Heart Murmurs
Mayo_clinic
Risk factors Things that increase the risk of heart murmurs in babies includes: Family history of heart problems linked to murmurs Uncontrolled diabetes in the mother during pregnancy German measles (rubella) in the mother during pregnancy Use of certain medications, alcohol or illegal drugs by the mother during pregnancy Some medical conditions can increase the risk of heart murmurs, including: A rare cancerous tumor that releases certain chemicals into the bloodstream (carcinoid syndrome) A weakened heart muscle (cardiomyopathy) An infection of the lining of the heart (endocarditis) Anemia Blood disorders marked by a high number of certain white cells, called eosinophils (hypereosinophilic syndrome) Certain autoimmune disorders such as lupus and rheumatoid arthritis Heart valve disease High blood pressure in the lungs (pulmonary hypertension) History of rheumatic fever Overactive thyroid (hyperthyroidism) Prevention There's no known prevention for heart murmurs.
-
Bronchiolitis
Mayo_clinic
Aspirin is not recommended in children due to the risk of Reye's syndrome, a rare but potentially life-threatening condition. Children and teenagers recovering from chickenpox or flu-like symptoms should never take aspirin, as they have a higher risk of Reye's syndrome. Avoid secondhand smoke. Smoke can worsen symptoms of respiratory infections.SLC35C1, TAP2, CXCL8, CCL5, SDCCAG8, PLCG2, PLEC, IL10, TAPBP, TAP1, TLR4, IFNG, VDR, CD14, TLR1, TLR10, IL4, TLR2, IL9, SFTPD, NOS2, IL13, IL17A, TNF, SFTPA1, IL5, COPD, IL18, JUN, ISYNA1, TLR9, HMGB1, IFNL3, CENPJ, CCL2, PEA15, NR1I2, ADRB2, SCGB1A1, TNFRSF1A, TLR3, PRDX2, SLC2A1, IL1RL1, TLR6, PPIP5K1, ABCB6, PLF, MIR27A, IFNL1, PGAM5, IFNLR1, ORMDL3, IL33, TSLP, SEMA4A, MAVS, TLR7, HPGDS, KLRK1, PTGDR2, RAPGEF4, SLC27A4, POSTN, BET1, ATP6AP2, CX3CL1, PTGDS, CCL4, FUT2, IFNA13, IFNA5, IFNA1, IFIT1, IFI27, ICAM1, GSTT1, GSTP1, NR3C1, GC, DHPS, CCL3, CX3CR1, CTLA4, CRP, COL5A1, CCR5, CLC, CD38, CD19, RUNX3, CAMP, IL2, IL3, IL4R, IL6, RNASE3, RNASE2, MOK, NECTIN1, AGER, PTGDR, PRF1, PMCH, SERPINF2, SERPINE1, OCA2, NFKBIA, MUC5AC, MBL2, LTA, LAIR1, IRF3, IL15, IL10RB, CXCR2, IL7, KLRC4-KLRK1
-
Aluminium Phosphide Poisoning
Wikipedia
The dominant clinical feature is severe hypotension refractory to dopamine therapy. [5] Other features may include dizziness, fatigue, tightness in the chest, headache, nausea , vomiting, diarrhoea , ataxia , numbness, paraesthesia , tremor, muscle weakness, diplopia and jaundice . [6] [7] [8] [9] If severe inhalation occurs, the patient may develop acute respiratory distress syndrome (ARDS), heart failure , arrhythmias , convulsion and coma . ... PMID 30078026 . v t e Poisoning Toxicity Overdose History of poison Inorganic Metals Toxic metals Beryllium Cadmium Lead Mercury Nickel Silver Thallium Tin Dietary minerals Chromium Cobalt Copper Iron Manganese Zinc Metalloids Arsenic Nonmetals Sulfuric acid Selenium Chlorine Fluoride Organic Phosphorus Pesticides Aluminium phosphide Organophosphates Nitrogen Cyanide Nicotine Nitrogen dioxide poisoning CHO alcohol Ethanol Ethylene glycol Methanol Carbon monoxide Oxygen Toluene Pharmaceutical Drug overdoses Nervous Anticholinesterase Aspirin Barbiturates Benzodiazepines Cocaine Lithium Opioids Paracetamol Tricyclic antidepressants Cardiovascular Digoxin Dipyridamole Vitamin poisoning Vitamin A Vitamin D Vitamin E Megavitamin-B 6 syndrome Biological 1 Fish / seafood Ciguatera Haff disease Ichthyoallyeinotoxism Scombroid Shellfish poisoning Amnesic Diarrhetic Neurotoxic Paralytic Other vertebrates amphibian venom Batrachotoxin Bombesin Bufotenin Physalaemin birds / quail Coturnism snake venom Alpha-Bungarotoxin Ancrod Batroxobin Arthropods Arthropod bites and stings bee sting / bee venom Apamin Melittin scorpion venom Charybdotoxin spider venom Latrotoxin / Latrodectism Loxoscelism tick paralysis Plants / fungi Cinchonism Ergotism Lathyrism Locoism Mushrooms Strychnine 1 including venoms , toxins , foodborne illnesses .
-
Gastroschisis
Wikipedia
Gastroschisis Gastrochisis in ultrasound: defect is right to midline Pronunciation gas-troh-skee-sis [1] Specialty General surgery , medical genetics Symptoms Intestines extend outside of the body through a hole next to the belly button [2] Complications Feeding problems, prematurity , intrauterine growth restriction [1] [3] Usual onset During early development [1] Causes Unknown [1] Risk factors Mother who smokes, drinks alcohol, or is younger than 20 [3] [1] Diagnostic method Ultrasound during pregnancy, based on symptom at birth [1] Differential diagnosis Omphalocele , prune belly syndrome [3] [4] Treatment Early surgery [1] Frequency 4 per 10,000 births [3] Gastroschisis is a birth defect in which the baby's intestines extend outside of the abdomen through a hole next to the belly button . [2] The size of the hole is variable, and other organs including the stomach and liver may also occur outside the baby's body. [1] Complications may include feeding problems, prematurity , intestinal atresia , and intrauterine growth restriction . [1] [3] The cause is typically unknown. [3] [1] Rates are higher in babies born to mothers who smoke, drink alcohol, or are younger than 20 years old. [3] [1] Ultrasounds during pregnancy may make the diagnosis. [1] Otherwise diagnosis occurs at birth. [1] It differs from omphalocele in that there is no covering membrane over the intestines. [3] Treatment involves surgery. [1] This typically occurs shortly after birth. [1] In those with large defects the exposed organs may be covered with a special material and slowly moved back into the abdomen. [1] The condition affects about 4 per 10,000 newborns. [3] Rates of the condition appear to be increasing. [1] Contents 1 Signs and symptoms 2 Causes 3 Pathophysiology 4 Diagnosis 5 Treatment 6 Prognosis 7 Epidemiology 8 See also 9 References 10 External links Signs and symptoms [ edit ] There are no signs during pregnancy. ... External links [ edit ] Classification D ICD - 10 : Q79.3 ICD - 9-CM : 756.73 OMIM : 230750 MeSH : D020139 DiseasesDB : 31155 SNOMED CT : 72951007 External resources MedlinePlus : 000992 eMedicine : ped/1642 radio/303 Orphanet : 2368 v t e Congenital diaphragm and abdominal wall defects, abdominopelvic cavity Thoracic diaphragm Hernia Congenital diaphragmatic hernia Bochdalek hernia Abdominal wall Omphalocele Gastroschisis Prune belly syndrome
-
Leukoencephalopathy, Hereditary Diffuse, With Spheroids
Omim
Two years later, he had a flat affect, inappropriate behavior, poor concentration, executive dysfunction, restless legs syndrome, and insomnia. There was psychomotor slowing, and ideomotor and constructional apraxia. ... Antemortem clinical diagnoses in mutation carriers included frontotemporal dementia (FTD; 600275), corticobasal syndrome, Alzheimer disease (AD; 104300), multiple sclerosis (MS; 126200), atypical cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL; 125310), and Parkinson disease (PD; 168600).